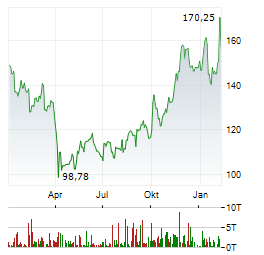
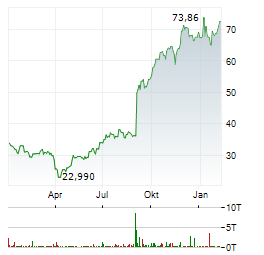
Chart-Typ:
Zeitraum:
| Community Rückblick: wO User zu Todesfall bei Newron Studie: "...der Rekrutierungsstop ist Formsache" | © Foto: Newron Pharmaceuticals SpAFDA-Stopp, ein Todesfall im klinischen Studienprogramm und große Hoffnungen auf den Schizophrenie-Wirkstoff Evenamide: Newron sorgt wieder für Debatten. Zwischen Zuversicht... ► Artikel lesen |
| ROUNDUP/Aktien New York Schluss: Gewinnmitnahmen stürzen Nasdaq in Turbulenzen | NEW YORK (dpa-AFX) - Die am Mittwoch schon eingeleitete Gewinnmitnahmen-Welle bei den heißgelaufenen US-Tech-Aktien hat zu Wochenschluss ein lange nicht gesehenes Ausmaß angenommen. Befeuert wurde... ► Artikel lesen |
| Aktien New York: Gewinnmitnahmen an Nasdaq verstärken sich deutlich | NEW YORK (dpa-AFX) - Die am Mittwoch schon eingeleitete Gewinnmitnahmen-Welle bei den heißgelaufenen US-Tech-Aktien hat zu Wochenschluss nochmals deutlich zugenommen. Als Belastung kam am Freitag hinzu... ► Artikel lesen |
| Biogen's Salanersen Wins FDA Breakthrough Therapy Designation For Spinal Muscular Atrophy Treatment | WESTON (dpa-AFX) - Thursday, Biogen Inc. (BIIB) announced that the U.S. Food and Drug Administration has granted salanersen Breakthrough Therapy Designation for the treatment of spinal muscular... ► Artikel lesen |
| Biogen Inc.: Biogen's Salanersen Receives FDA Breakthrough Therapy Designation for Spinal Muscular Atrophy | Designation is supported by an exploratory analysis from the Phase 1b study showing that some children with SMA previously treated with gene therapy who had suboptimal clinical status experienced slowing... ► Artikel lesen |
| Biogen Inc.: EULAR 2026: Dapirolizumab Pegol Shows Potential to Reduce Flare Rates and Maintain Disease Control in Systemic Lupus Erythematosus | Steroid use reduction and disease control: In additional results from the Phase 3 PHOENYCS GO study, dapirolizumab pegol (DZP) plus standard of care was associated with sustained disease control at... ► Artikel lesen |
| XETR NEW INSTRUMENTS AVAILABLE ON XETRA - 04.06.2026 | Folgende Wertpapiere werden in den Handel an folgenden Handelsplätzen (MIC Code) im Quotation Board an der Frankfurter Wertpapierbörse (FWB) aufgenommen. Die Notierung an anderen Handelsplätzen im Quotation... ► Artikel lesen |
| EU Approves DAWNZERA For Hereditary Angioedema, Says Ionis And Otsuka | TOKYO (dpa-AFX) - Ionis Pharmaceuticals, Inc. (IONS) and Otsuka Pharmaceutical Co., Ltd. on Wednesday said the European Commission has approved Dawnzera to prevent repeated attacks of hereditary... ► Artikel lesen |
| Ionis Pharmaceuticals, Inc.: DAWNZERA (donidalorsen) approved in the European Union for hereditary angioedema (HAE) | Ionis Pharmaceuticals, Inc. (Nasdaq: IONS) and Otsuka Pharmaceutical Co., Ltd. (Otsuka) today announced that the European Commission (EC) has approved DAWNZERA (donidalorsen) in the European Union... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu BG MEDICINE INC finden Sie auf Wallstreet Online.