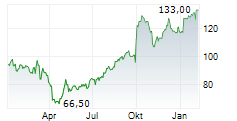
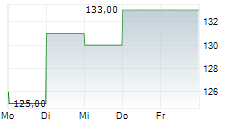
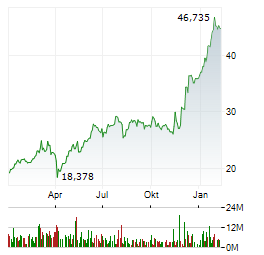
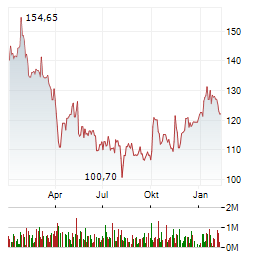
Chart-Typ:
Zeitraum:
| DAX beinahe regungslos - Infineon weiter gefragt, SAP und Bayer im Minus | Viel bewegt sich nicht in diesen Tagen im DAX. Ein Teil der Anleger schaut weiter in Richtung Wall Street und lässt sich von den beinahe täglichen Indexrekorden anstecken. Im Vergleich zu den US-Aktien... ► Artikel lesen |
| Bayer Aktie: Neue Rechtsklagen überlagern die Fortschritte | Frische Rechtsschlagzeilen aus den USA belasten die Bayer Aktie erneut. Zwei neue Fälle sorgen für Unruhe: Die Anfechtung des Roundup-Vergleichs wirft Fragen zum wichtigsten Entlastungsmeilenstein auf... ► Artikel lesen |
| Aktie trotzdem mit 50%-Chance?: Bayer unter Druck: Neuer Glyphosat-Richter macht Anleger nervös | © Foto: Patrick Pleul - dpa-ZentralbildEin US-Bundesrichter, der Bayers Sammelklage-Vergleich schon 2020 ablehnte, übernimmt nun erneut das Steuer im Glyphosat-Verfahren. Die Aktie reagiert mit deutlichen... ► Artikel lesen |
| JPMORGAN stuft MERCK KGAA auf 'Overweight' | NEW YORK (dpa-AFX Analyser) - Die US-Bank JPMorgan hat die Einstufung für Merck KGaA mit einem Kursziel von 150 Euro auf "Overweight" belassen. Die Studiendaten von Varegacestat hätten zwar auf eine... ► Artikel lesen |
| Während Merck, Amgen, Novo Nordisk und Fresenius Milliarden bewegen, wächst die Fantasie im Zelltherapie-Sektor | Anzeige / Werbung
Der Biotech-Sektor beginnt sich spürbar zu verändern. Während
Anleger monatelang fast ausschließlich auf KI-Aktien und Halbleiterwerte blickten, fließt inzwischen wieder deutlich... ► Artikel lesen |
| MilliporeSigma's Approach to Embedding Sustainability Earlier in Product Development | NORTHAMPTON, MA / ACCESS Newswire / May 28, 2026 / For MilliporeSigma, the U.S. and Canada Life Science business of Merck KGaA, Darmstadt, Germany, sustainability isn't treated as a standalone initiative.... ► Artikel lesen |
| Börsenrotation ohne KI! Jetzt 100 % mit MustGrow, Bayer und Novo Nordisk, Achtung bei K+S | Erstellt und veröffentlicht im Auftrag von MustGrow Biologics Corp. Perspektivenwechsel bleibt das Gebot der Stunde. Denn gestern tagte die EZB zur aktuellen Lage bei Inflation und Zinsniveau. Mit einer... ► Artikel lesen |
| Novo Nordisk: Wegovy-Pille hinkt hinterher, FDA-Druck wächst | Zwei Analystenteams haben sich in diesen Tagen zur Novo Nordisk Aktie geäußert, beide mit neutralen Einstufungen. Im Mittelpunkt stehen die Marktdurchdringung der oralen Wegovy-Version und das regulatorische... ► Artikel lesen |
| JEFFERIES stuft NOVO NORDISK auf 'Hold' | NEW YORK (dpa-AFX Analyser) - Das Analysehaus Jefferies hat die Einstufung für Novo Nordisk nach einem Gespräch mit einem Rechtsexperten zum Thema GLP-1-Wirkstoffe auf "Hold" mit einem Kursziel von... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu UCB SA ADR finden Sie auf Wallstreet Online.