| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
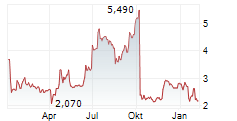
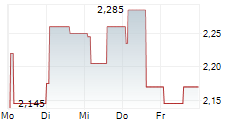
| 2,155 | 2,445 | 20:22 |
Chart-Typ:
Zeitraum:
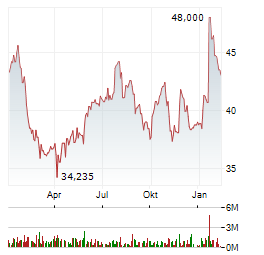
| DEUTSCHE BANK RESEARCH stuft QIAGEN NV auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat Qiagen mit einem Kursziel von 43 US-Dollar auf "Buy" belassen. Das Feedback aus Gesprächen mit etlichen US-Laborexperten deute darauf hin, dass... ► Artikel lesen |
| QIAGEN N.V.: QIAGEN unterstützt Forschungs- und Überwachungsmaßnahmen zum Ausbruch des Bundibugyo-Ebolavirus | Ab sofort stehen zwei maßgeschneiderte QIAcuity digitale PCR-Assays für den Einsatz in der Forschung (Research Use Only, RUO) zur Verfügung, die auf die mit dem aktuellen Ausbruch in Verbindung... ► Artikel lesen |
| QIAGEN N.V.: QIAGEN Supports Infectious Disease Research and Surveillance Efforts for Bundibugyo Ebolavirus Outbreak | Two custom-designed research-use-only (RUO) QIAcuity digital PCR assays now available to target Bundibugyo ebolavirus sequences associated with the current outbreak Assays designed for... ► Artikel lesen |
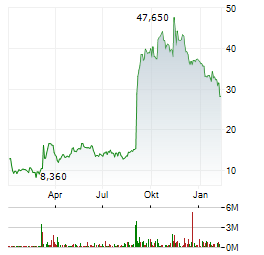
| Mineralys Therapeutics, Inc.: Mineralys Therapeutics Announces Repurchase of Royalty Obligations in Tanabe License Agreement and Concurrent Financing | - Repurchase of lorundrostat royalty obligation in Tanabe license agreement for $200 million upfront and up to $100 million once certain commercial milestones are met - - $500 million committed... ► Artikel lesen |
| Mineralys Therapeutics, Inc.: Mineralys Therapeutics Presents New Data from the Phase 3 Launch-HTN Trial of Lorundrostat in Participants with Hypertension and Chronic Kidney Disease at European Meeting on Hypertension and Cardiovascular Protection ... | - Post hoc analysis from pivotal Launch-HTN trial shows statistically significant and clinically meaningful reductions in blood pressure in participants with chronic kidney disease - - In participants... ► Artikel lesen |
| Mineralys Therapeutics, Inc.: Mineralys Therapeutics Reports First Quarter 2026 Financial Results and Provides Corporate Update | - NDA for lorundrostat in adults with hypertension accepted by FDA; assigned PDUFA target date of December 22, 2026 - - Conference call today at 4:30 p.m. ET - RADNOR, Pa., May 06, 2026 (GLOBE NEWSWIRE)... ► Artikel lesen |
| Praxis Precision Medicines, Inc.: Praxis Precision Medicines Provides Vormatrigine Program Update | POWER1 Study in highly refractory patients with focal onset seizures did not meet its primary success measure Secondary measure, the 50% response rate, was met and seizure reduction during the second... ► Artikel lesen |
| Praxis Precision Medicines, Inc.: Praxis Precision Medicines Provides Corporate Update and Reports First Quarter 2026 Financial Results | FDA accepted the new drug application (NDA) for ulixacaltamide in Essential Tremor with a PDUFA target action date of January 29, 2027, and the NDA for relutrigine, with priority review, in SCN2A... ► Artikel lesen |
| Praxis Precision Medicines, Inc.: Praxis Precision Medicines Announces FDA Acceptance of New Drug Application for Ulixacaltamide HCl in Patients with Essential Tremor | FDA assigned PDUFA target action date of January 29, 2027 No advisory committee meeting expected BOSTON, April 14, 2026 (GLOBE NEWSWIRE) -- Praxis Precision Medicines, Inc. (NASDAQ: PRAX), a fully... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu XSPRAY PHARMA AB finden Sie auf Wallstreet Online.