| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 2,165 | 2,370 | 22.05. |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 12.05. | XSpray Pharma AB: Bulletin from the annual general meeting of Xspray Pharma AB (publ) 12 May 2026 | 113 | GlobeNewswire (Europe) | The following resolutions were passed at the annual general meeting (the "AGM") of Xspray Pharma AB (publ) ("Xspray") on 12 May 2026.Adoption of income statement and balance sheet for the financial... ► Artikel lesen | |
| 28.04. | XSpray Pharma AB: Interim Report First Quarter 2026 | 174 | GlobeNewswire (Europe) | January-March 2026, GroupNet sales amounted to SEK 0 thousand (0)Earnings before tax amounted to SEK -35,380 thousand (-42,321)Earnings per share before dilution amounted to SEK -0.85 (-1.14)Cash flow... ► Artikel lesen | |
| XSPRAY PHARMA Aktie jetzt für 0€ handeln | |||||
| 23.04. | XSpray Pharma AB: Xspray's rights issue oversubscribed - over-allotment issue fully utilized | 137 | GlobeNewswire (Europe) | THIS PRESS RELEASE MAY NOT BE MADE PUBLIC, PUBLISHED OR DISTRIBUTED, DIRECTLY OR INDIRECTLY, IN OR INTO THE UNITED STATES OF AMERICA, AUSTRALIA, BELARUS, CANADA, HONG KONG, JAPAN, NEW ZEALAND, RUSSIA... ► Artikel lesen | |
| 14.04. | XSpray Pharma AB: FDA accepts trade name Nilopki for Xspray's drug candidate XS003 (nilotinib) | 1.990 | GlobeNewswire (Europe) | The US Food and Drug Administration (FDA) has accepted Nilopki® as the proprietary name for Xspray Pharma's drug candidate XS003 (nilotinib). Nilopki is an improved, amorphous formulation of nilotinib... ► Artikel lesen | |
| 31.03. | XSpray Pharma AB: Xspray publishes disclosure document regarding rights issue | 141 | GlobeNewswire (Europe) | THIS PRESS RELEASE MAY NOT BE MADE PUBLIC, PUBLISHED OR DISTRIBUTED, DIRECTLY OR INDIRECTLY, IN OR INTO THE UNITED STATES OF AMERICA, AUSTRALIA, BELARUS, CANADA, HONG KONG, JAPAN, NEW ZEALAND, RUSSIA... ► Artikel lesen | |
| 31.03. | XFRA CAPITAL ADJUSTMENT INFORMATION - 31.03.2026 | 297 | Xetra Newsboard | Das Instrument OJ3 SE0009779796 SEAFIRE CAPITAL SK 10 EQUITY wird ex Kapitalmassnahme gehandelt am 31.03.2026 The instrument OJ3 SE0009779796 SEAFIRE CAPITAL SK 10 EQUITY is traded ex capital adjustment... ► Artikel lesen | |
| 26.03. | XSpray Pharma AB: Xspray receives additional subscription undertakings; rights issue now covered to 110% and over-allotment issue increased | 212 | GlobeNewswire (Europe) | THIS PRESS RELEASE MAY NOT BE MADE PUBLIC, PUBLISHED OR DISTRIBUTED, DIRECTLY OR INDIRECTLY, IN OR INTO THE UNITED STATES OF AMERICA, AUSTRALIA, BELARUS, CANADA, HONG KONG, JAPAN, NEW ZEALAND, RUSSIA... ► Artikel lesen | |
| 24.03. | XSpray Pharma AB: Xspray resolves on a rights issue of approximately SEK 83 million with an over-allotment issue | 188 | GlobeNewswire (Europe) | THIS PRESS RELEASE MAY NOT BE MADE PUBLIC, PUBLISHED OR DISTRIBUTED, DIRECTLY OR INDIRECTLY, IN OR INTO THE UNITED STATES OF AMERICA, AUSTRALIA, BELARUS, CANADA, HONG KONG, JAPAN, NEW ZEALAND, RUSSIA... ► Artikel lesen | |
| 11.03. | XSpray Pharma AB: Xspray Pharma: FDA accepts resubmitted application for market approval of Dasynoc - PDUFA date August 25, 2026 | 333 | GlobeNewswire (Europe) | The U.S. Food and Drug Administration (FDA) has accepted Xspray Pharma's resubmitted New Drug Application (NDA) for Dasynoc® for review under the 505(b)(2) procedure. The FDA has set a PDUFA date of... ► Artikel lesen | |
| 26.02. | XSpray Pharma AB: Xspray Pharma re-submits its FDA application for Dasynoc | 253 | GlobeNewswire (Europe) | Xspray Pharma AB (publ) has re-submitted its application for market approval for Dasynoc® to the U.S. Food and Drug Administration (FDA). Dasynoc® is the company's lead product candidate - an amorphous... ► Artikel lesen | |
| 26.02. | XSpray Pharma AB: Xspray Pharma appoints Blake Leitch as CEO - Per Andersson continues as Chief Scientific Officer (CSO) | 274 | GlobeNewswire (Europe) | The Board of Directors of Xspray Pharma has today appointed Blake Leitch as CEO of Xspray Pharma, effective no later than June 1, 2026. He brings over 20 years of international experience of marketing... ► Artikel lesen | |
| 12.02. | XSpray Pharma AB: Interim Report Fourth Quarter 2025 | 147 | GlobeNewswire (Europe) | October-December 2025, GroupNet sales amounted to SEK 0 thousand (0)Earnings before tax amounted to SEK -37,009 thousand (-82,002)Earnings per share before dilution amounted to SEK -0.89 (-2.36)Cash... ► Artikel lesen | |
| 05.11.25 | XSpray Pharma AB: Interim Report Third Quarter 2025 | 280 | GlobeNewswire (Europe) | July-September 2025, GroupNet sales amounted to SEK 0 thousand (0)Earnings before tax amounted to SEK -47,332 thousand (-82,272)Earnings per share before dilution amounted to SEK -1.25 (-2.44)Cash flow... ► Artikel lesen | |
| 08.10.25 | XSpray Pharma AB: Xspray Pharma provides update on the FDA process for Dasynoc - observations at contract manufacturer delay approval | 316 | GlobeNewswire (Europe) | • FDA has issued a Complete Response Letter (CRL) regarding Xspray Pharma's New Drug Application (NDA) for Dasynoc, referring to GMP (Good Manufacturing Practice) observations at a contract manufacturer.... ► Artikel lesen | |
| 11.09.25 | XSpray Pharma AB: Xspray's rights issue oversubscribed - over-allotment issue increased and fully utilized | 345 | GlobeNewswire (Europe) | THIS PRESS RELEASE MAY NOT BE MADE PUBLIC, PUBLISHED OR DISTRIBUTED, DIRECTLY OR INDIRECTLY, IN OR INTO THE UNITED STATES OF AMERICA, AUSTRALIA, BELARUS, CANADA, HONG KONG, JAPAN, NEW ZEELAND, RUSSIA... ► Artikel lesen | |
| 26.08.25 | Listing of subscription rights and paid subscription shares of XSpray Pharma AB | 446 | GlobeNewswire | With effect from August 27, 2025, the subscription rights of XSpray Pharma AB will be traded on the list for Equity rights. Trading will continue up until and including September 04, 2025.
Instrument:
Subscription... ► Artikel lesen | |
| 25.08.25 | XSpray Pharma AB: Xspray publishes disclosure document regarding rights issue | 462 | GlobeNewswire (Europe) | THIS PRESS RELEASE MAY NOT BE MADE PUBLIC, PUBLISHED OR DISTRIBUTED, DIRECTLY OR INDIRECTLY, IN OR INTO THE UNITED STATES OF AMERICA, AUSTRALIA, BELARUS, CANADA, HONG KONG, JAPAN, NEW ZEELAND, RUSSIA... ► Artikel lesen | |
| 21.08.25 | XFRA CAPITAL ADJUSTMENT INFORMATION - 21.08.2025 | 780 | Xetra Newsboard | Das Instrument 6O1 CA13810W1023 CANTER RESOURCES CORP. EQUITY wird cum Kapitalmassnahme gehandelt am 21.08.2025 und ex Kapitalmassnahme am 22.08.2025 The instrument 6O1 CA13810W1023 CANTER RESOURCES... ► Artikel lesen | |
| 19.08.25 | XSpray Pharma AB: Xspray Pharma Submits XS003 to the FDA - The Company's Second Product Candidate from the HyNap Platform | 572 | GlobeNewswire (Europe) | Xspray Pharma (Nasdaq Stockholm: XSPRAY) has submitted a New Drug Application (NDA) to the U.S. Food and Drug Administration (FDA) for its product candidate XS003 (nilotinib) for the treatment of chronic... ► Artikel lesen | |
| 15.08.25 | XSpray Pharma AB: Interim Report Second Quarter 2025 | 330 | GlobeNewswire (Europe) | April-June 2025, GroupNet sales amounted to SEK 0 thousand (0)Earnings before tax amounted to SEK -44,885 thousand (-53,620)Earnings per share before dilution amounted to SEK -2.35 (-1.64)Cash flow... ► Artikel lesen |
1 2 Weiter >>
23 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| SUMMIT THERAPEUTICS | 16,620 | 0,00 % | Cantor Fitzgerald reiterates Overweight on Summit Therapeutics stock | ||
| CG ONCOLOGY | 64,25 | -0,53 % | CG Oncology Inc.: CG Oncology Reports Positive First Results from CORE-008 Cohort CX Phase 2 Trial Evaluating Intravesical Combination Therapy in High-Risk BCG-Exposed and BCG-Unresponsive Patients | - High CR rates at any time observed in the CIS-containing population with 85.7% and 92.3% in the ITT population and Efficacy Evaluable population, respectively - High-Grade - EFS in the overall intention-to-treat... ► Artikel lesen | |
| QIAGEN | 30,290 | +0,65 % | QIAGEN N.V.: QIAGEN to advance AI-driven drug discovery with graph-based AI and curated bioinformatics knowledge with NVIDIA | Collaboration integrates NVIDIA accelerated computing and the NVIDIA BioNeMo platform with QIAGEN Digital Insights' curated biomedical knowledge bases Goal to advance how disease mechanisms... ► Artikel lesen | |
| TANGO THERAPEUTICS | 20,240 | -6,38 % | Tango Therapeutics Shares Fall 17% | ||
| PRAXIS PRECISION MEDICINES | 343,26 | 0,00 % | Praxis Precision Medicines, Inc.: Praxis Precision Medicines Provides Corporate Update and Reports First Quarter 2026 Financial Results | FDA accepted the new drug application (NDA) for ulixacaltamide in Essential Tremor with a PDUFA target action date of January 29, 2027, and the NDA for relutrigine, with priority review, in SCN2A... ► Artikel lesen | |
| COGENT BIOSCIENCES | 32,640 | +0,18 % | Cogent Biosciences: Aktie legt vor wichtigen Studiendaten um 4 % zu | ||
| GEOVAX LABS | 3,630 | 0,00 % | EQS-News: GeoVax, Inc.: GeoVax Comments on Escalating Bundibugyo Ebola Outbreak and Growing Need for Flexible Biodefense Vaccine Platforms | EQS-News: GeoVax, Inc.
/ Key word(s): Science
GeoVax Comments on Escalating Bundibugyo Ebola Outbreak and Growing Need for Flexible Biodefense Vaccine Platforms 20.05.2026 / 16:06... ► Artikel lesen | |
| METAVIA | 3,850 | 0,00 % | MetaVia: Präklinische Studie zu Vanoglipel untermauert Potenzial bei Lebererkrankungen | ||
| IMMUNEERING | 5,390 | +3,26 % | Immuneering Corporation: Immuneering Reports 17.3 Months Median Overall Survival in First-Line Metastatic Pancreatic Cancer Patients Treated with Atebimetinib Plus Chemotherapy | - Detailed data from 55 patients to be shared in an oral presentation at the ASCO Annual Meeting on June 1, 2026 - - Tolerability profile consistent with prior updates: only two categories of Grade... ► Artikel lesen | |
| RELAY THERAPEUTICS | 13,535 | -0,92 % | Relay Therapeutics, Inc.: Relay Therapeutics Announces Pricing of Public Offering of Common Stock | CAMBRIDGE, Mass., May 20, 2026 (GLOBE NEWSWIRE) -- Relay Therapeutics, Inc. (Nasdaq: RLAY), a clinical-stage, small molecule precision medicine company developing potentially life-changing therapies... ► Artikel lesen | |
| TAYSHA GENE THERAPIES | 5,490 | -0,90 % | Taysha Gene Therapies, Inc.: Taysha Gene Therapies Reports First Quarter 2026 Financial Results and Provides Corporate Update | Reaffirmed FDA alignment on TSHA-102 BLA pathway, including pivotal trial design, endpoints and potential to submit for approval based on REVEAL pivotal trial 6-month interim analysis, following... ► Artikel lesen | |
| IMMUNOVANT | 34,140 | 0,00 % | Immunovant Inc.: Immunovant Provides Corporate Updates and Reports Financial Results for the Fourth Quarter and Fiscal Year Ended March 31, 2026 | IMVT-1402 showed clinically meaningful response rates of 72.7% ACR20, 54.5% ACR50 and 35.8% ACR70 at Week 16 in the open label period of its trial in difficult-to-treat rheumatoid arthritis (D2T RA)... ► Artikel lesen | |
| BEAM THERAPEUTICS | 28,640 | +3,51 % | Beam Therapeutics presents AATD gene therapy trial data at ATS | ||
| INHIBRX BIOSCIENCES | 101,45 | -8,25 % | Inhibrx Biosciences, Inc.: Inhibrx Reports First Quarter 2026 Financial Results | SAN DIEGO, May 14, 2026 /PRNewswire/ -- Inhibrx Biosciences, Inc. (Nasdaq: INBX) ("Inhibrx" or the "Company") today reported financial results for the first quarter... ► Artikel lesen | |
| RECURSION PHARMACEUTICALS | 3,005 | 0,00 % | Recursion Pharmaceuticals: Recursion Reports First Quarter Financial Results and Provides Business Update | Multiple milestones achieved or on track across wholly owned and partnered programs REC-1245 (RBM39 degrader): Early clinical data in solid tumors demonstrate a well-tolerated safety profile and predictable... ► Artikel lesen |
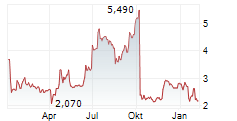
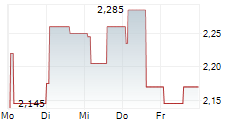
XSPRAY PHARMA AB gehört der Branche Biotechnologie an. Der aktuelle Kurs liegt bei 2,180 Euro und damit -1,58 % im Minus.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu XSPRAY PHARMA AB finden Sie auf Wallstreet Online.