- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| ENLIVEN THERAPEUTICS Aktie jetzt für 0€ handeln | |||||
| Mo | Stifel initiates Enliven Therapeutics stock with buy rating | 1 | Investing.com | ||
| Mo | Stifel startet Coverage für Enliven Therapeutics mit 'Buy'-Rating | 2 | Investing.com Deutsch | ||
| 03.06. | Enliven to host webcast on ELVN-001 trial data June 11 | 1 | Investing.com | ||
| 03.06. | Enliven Therapeutics: Webcast zu neuen Daten der ELVN-001-Studie am 11. Juni | 2 | Investing.com Deutsch | ||
| 02.06. | Mizuho erhöht Kursziel für Enliven Therapeutics im Vorfeld von Studiendaten | 4 | Investing.com Deutsch | ||
| 02.06. | Mizuho raises Enliven Therapeutics stock price target on trial data | 1 | Investing.com | ||
| 12.05. | Enliven to present updated ELVN-001 leukemia trial data at EHA | 3 | Investing.com | ||
| 12.05. | Guggenheim initiates Enliven Therapeutics stock with buy rating | 2 | Investing.com | ||
| 11.05. | Why is Enliven Therapeutics (ELVN) One of the Best Performing Healthcare Stocks so Far in 2026? | 1 | Insider Monkey | ||
| 07.05. | Enliven Therapeutics, Inc.: Enliven Therapeutics Reports First Quarter Financial Results and Provides a Business Update | 247 | PR Newswire | Phase 1 data update for ELVN-001 expected mid-2026
Initiation of the Phase 3 ENABLE-2 pivotal trial of ELVN-001 expected in the second half of 2026
Strong balance... ► Artikel lesen | |
| 07.05. | Enliven Therapeutics, Inc. - 8-K, Current Report | - | SEC Filings | ||
| 17.04. | Enliven Therapeutics (ELVN) Up 194% This Year on Strong CML Market | 3 | Insider Monkey | ||
| 08.04. | H.C. Wainwright reiterates Enliven Therapeutics stock rating on Terns deal details | 5 | Investing.com | ||
| 26.03. | Enliven Therapeutics stock price target raised to $56 by H.C. Wainwright | 1 | Investing.com | ||
| 26.03. | Enliven Therapeutics (ELVN) Soars to 5-Year High as Analysts 'Upbeat' | 4 | Insider Monkey | ||
| 25.03. | Clear Street reiterates Enliven Therapeutics stock Buy on CML opportunity | 2 | Investing.com | ||
| 24.03. | Mizuho hebt Kursziel für Enliven Therapeutics nach CML-Daten auf 45 US-Dollar an | 4 | Investing.com Deutsch | ||
| 24.03. | Mizuho raises Enliven Therapeutics price target to $45 on CML data | 5 | Investing.com | ||
| 03.03. | Enliven Therapeutics, Inc. - S-8, Securities to be offered to employees in employee benefit plans | 1 | SEC Filings | ||
| 03.03. | Enliven Therapeutics: Q4 Earnings Insights | 1 | Benzinga.com |
1 2 Weiter >>
34 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 74,80 | -0,20 % | Rendite-Chance Krebsmarkt: Mit BioNTech, Vidac Pharma und Pfizer den nächsten Mrd.-Trigger erwischen | Die Alterung der Gesellschaft lässt die Gesundheitsausgaben rasant steigen. Im Krebssegment zeichnet sich ein Billionenmarkt ab, denn bis 2040 werden jährlich über 30 Mio. Neuerkrankungen erwartet.... ► Artikel lesen | |
| EVOTEC | 4,750 | -3,69 % | Aktien - Shortseller-Positionen: Carl Zeiss Meditec, Evotec, HelloFresh, Kontron, LPKF Laser, TeamViewer | Wer Aktien leerverkauft, also auf fallende Kurse spekuliert (Shortselling), unterliegt bestimmten Transparenzpflichten. Aktuelle Meldungen über Leerverkäufe geben Aufschluss darüber. Diese Vorgaben... ► Artikel lesen | |
| BB BIOTECH | 44,950 | -0,77 % | Aktien KW 17 Märkte im Würgegriff. Planloser Trump , selbstbewusterer Iran. Lösungen müssen her. News. Rheinmetall. ITM Power. Porsche. The Platform Group. K+S. Bugatti. Circus. Surteco. NanoRepro. BB Biotech. Mensch und Maschine. ATOSS. MHP Hotel | Aktien - letzte Woche warteten die Märkte auf eine Lösung des Iran-Kriegs. Aber die Iraner sehen sich im Vorteil und Trump verlängert "auf unbestimmte Zeit" den Waffenstillstand. Aber die Strasse von... ► Artikel lesen | |
| QIAGEN | 31,740 | -0,02 % | QIAGEN NV - Stabilität als Ausgangspunkt für Struktur | ||
| MODERNA | 41,295 | -0,21 % | Neue CAR-T-Erfolge bei Autoimmunerkrankungen: Warum Lösungen für Zelltherapien vor dem nächsten Wachstumsschub stehen könnten | ||
| VALNEVA | 2,270 | -0,83 % | EILMELDUNG bei Valneva: Der Kurs nähert sich einer gefährlichen Marke - So reagieren erfahrene Anleger jetzt | ||
| AMGEN | 299,30 | +0,25 % | EQS-News: Amgen: Europäische Kommission erteilt Zulassung für Imdylltra von Amgen für die Behandlung von kleinzelligem Lungenkrebs im fortgeschrittenen Stadium | EQS-News: Amgen
/ Schlagwort(e): Sonstiges/Sonstiges
EUROPÄISCHE KOMMISSION ERTEILT ZULASSUNG FÜR IMDYLLTRA VON AMGEN FÜR DIE BEHANDLUNG VON KLEINZELLIGEM... ► Artikel lesen | |
| NOVAVAX | 7,550 | +0,40 % | Novavax set for a seventh straight decline | ||
| BIOGEN | 171,52 | -0,58 % | Biogen's Salanersen Wins FDA Breakthrough Therapy Designation For Spinal Muscular Atrophy Treatment | WESTON (dpa-AFX) - Thursday, Biogen Inc. (BIIB) announced that the U.S. Food and Drug Administration has granted salanersen Breakthrough Therapy Designation for the treatment of spinal muscular... ► Artikel lesen | |
| BIOFRONTERA | 2,600 | +1,56 % | PTA-News: Biofrontera AG: Biofrontera berichtet über die Ergebnisse des ersten Quartals 2026 | DJ PTA-News: Biofrontera AG: BIOFRONTERA BERICHTET ÜBER DIE ERGEBNISSE DES ERSTEN QUARTALS 2026
Unternehmensmitteilung für den Kapitalmarkt
Biofrontera AG: BIOFRONTERA BERICHTET ÜBER... ► Artikel lesen | |
| HEIDELBERG PHARMA | 2,710 | +3,83 % | HEIDELBERG PHARMA AG: Ruhe vor der nächsten Phase | ||
| CRISPR THERAPEUTICS | 44,725 | +0,24 % | CRISPR Therapeutics Has $2.4 Billion in Cash and an Approved Drug. Why Is Its Stock Trading Nearly 40% Below the Wall Street Consensus? | ||
| REGENERON PHARMACEUTICALS | 531,60 | -0,39 % | CytomX Therapeutics Inc.: CytomX Announces Expansion of Strategic Research Collaboration with Regeneron in the Field of Conditional Bispecific Therapeutics for the Treatment of Cancer | - Expanded collaboration builds upon research momentum in developing next-generation bispecific immunotherapies using CytomX's PROBODY- and Regeneron's Veloci-Bi- platforms - - CytomX to receive... ► Artikel lesen | |
| BIOXXMED | 0,800 | 0,00 % | PTA-News: bioXXmed AG: Vollzug des Verkaufs der Rancoderm GmbH | DJ PTA-News: bioXXmed AG: Vollzug des Verkaufs der Rancoderm GmbH
Unternehmensmitteilung für den Kapitalmarkt
bioXXmed AG: Vollzug des Verkaufs der Rancoderm GmbH
Heidelberg (pta000/09.03.2026/13:50... ► Artikel lesen | |
| OCUGEN | 1,062 | 0,00 % | Ocugen, Inc. - 8-K, Current Report |
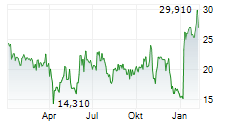
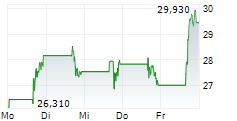
Das Unternehmen ENLIVEN THERAPEUTICS INC kann der Branche Biotechnologie zugeordnet werden. Die relative Kursveränderung beträgt aktuell +0,04 % bei einem Aktienkurs von 36,125 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu ENLIVEN THERAPEUTICS INC finden Sie auf Wallstreet Online.