| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 610,10 | 611,90 | 19:20 | |
| 610,10 | 611,90 | 19:17 |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| So | Mesoblast Aktie: Zelltherapie im Aufwind - warum der kleine Biotech-Player plötzlich in einer Liga mit Merck, Regeneron & Co. gedacht werden muss | Small- & Micro Cap Investment | |||
| Fr | Regeneron Pharmaceuticals, Inc.: Regeneron Named to Dow Jones Best-in-Class World Index for Global Responsibility Leadership for Seven Consecutive Years | 4 | GlobeNewswire (USA) | ||
| REGENERON PHARMACEUTICALS Aktie jetzt für 0€ handeln | |||||
| 05.05. | Regeneron Pharmaceuticals, Inc.: Dupixent (dupilumab) Demonstrates Improved Esophageal Function in Eosinophilic Esophagitis (EoE) Phase 4 Trial | 2 | GlobeNewswire (USA) | ||
| 02.05. | Is Regeneron a Buy After the FDA Approved its Gene Therapy to Restore Hearing? | 6 | The Motley Fool | ||
| 01.05. | Bristol Myers' legacy lift, Biogen's blue skies and Regeneron's murky outlook | 19 | BioPharma Dive | ||
| 30.04. | Regeneron Pharmaceuticals Analysts Cut Their Forecasts Following Q1 Earnings | 8 | Benzinga.com | ||
| 30.04. | Regeneron Q1 2026 net income falls 10% to $727m | 8 | Pharmaceutical Technology | ||
| 29.04. | Truist cuts Regeneron stock price target on FDA approval delay | 16 | Investing.com | ||
| 29.04. | Regeneron Flags FDA Delays For Eylea HD Prefilled Syringe | 4 | Benzinga.com | ||
| 29.04. | Raymond James cuts Regeneron stock price target on Eylea HD miss | 13 | Investing.com | ||
| 29.04. | Regeneron-Aktie fällt um 5 % nach enttäuschenden Eylea-Zahlen und regulatorischem Rückschlag | 29 | Investing.com Deutsch | ||
| 29.04. | Regeneron's quarterly sales of Eylea drop below $1B for the first time in 8 years | 25 | FiercePharma | ||
| 29.04. | Regeneron im ersten Quartal 2026: Dupixent treibt Umsatz um 19 % an | 10 | Investing.com Deutsch | ||
| 29.04. | Regeneron Q1 2026 slides: Dupixent drives 19% revenue growth | 7 | Investing.com | ||
| 29.04. | Regeneron drops despite Q1 beat, $3B share buyback program | 17 | Seeking Alpha | ||
| 29.04. | Regeneron (REGN) Surpasses Q1 Earnings and Revenue Estimates | 9 | Zacks | ||
| 29.04. | Regeneron Pharmaceuticals Inc Reveals Drop In Q1 Bottom Line | 282 | AFX News | WASHINGTON (dpa-AFX) - Regeneron Pharmaceuticals Inc (REGN) reported a profit for first quarter that Drops, from the same period last yearThe company's earnings totaled $727.2 million, or $6.75... ► Artikel lesen | |
| 29.04. | Regeneron Pharmaceuticals, Inc.: Regeneron Reports First Quarter 2026 Financial and Operating Results | 610 | GlobeNewswire (Europe) | First quarter 2026 revenues increased 19% to $3.6 billion versus first quarter 2025Dupixent- global net sales (recorded by Sanofi) increased 33% to $4.9 billion EYLEA HD- U.S. net sales increased... ► Artikel lesen | |
| 29.04. | Regeneron Pharmaceuticals Non-GAAP EPS of $9.47 beats by $0.57, revenue of $3.61B beats by $130M | 12 | Seeking Alpha | ||
| 29.04. | REGENERON PHARMACEUTICALS, INC. - 10-Q, Quarterly Report | 3 | SEC Filings |
1 2 3 4 5 Weiter >>
339 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 78,90 | -1,13 % | Curevac-Gründer wirft Biontech Täuschung vor | TÜBINGEN/MAINZ (dpa-AFX) - Nach der Übernahme des Biotechunternehmens Curevac durch Biontech wirft Curevac-Gründer Ingmar Hoerr dem Konkurrenten Täuschung vor. Hintergrund sind die Schließungspläne... ► Artikel lesen | |
| EVOTEC | 4,540 | -10,63 % | Umbaukosten drücken Evotec zum Jahresstart operativ ins Minus | HAMBURG (dpa-AFX) - Der Pharmawirkstoffforscher und -entwickler Evotec ist noch schwächer ins Jahr gestartet als befürchtet. Hohe Umbaukosten belasteten dabei das Ergebnis. Der bereinigte Gewinn vor... ► Artikel lesen | |
| BB BIOTECH | 50,000 | +0,60 % | Aktien KW 17 Märkte im Würgegriff. Planloser Trump , selbstbewusterer Iran. Lösungen müssen her. News. Rheinmetall. ITM Power. Porsche. The Platform Group. K+S. Bugatti. Circus. Surteco. NanoRepro. BB Biotech. Mensch und Maschine. ATOSS. MHP Hotel | Aktien - letzte Woche warteten die Märkte auf eine Lösung des Iran-Kriegs. Aber die Iraner sehen sich im Vorteil und Trump verlängert "auf unbestimmte Zeit" den Waffenstillstand. Aber die Strasse von... ► Artikel lesen | |
| MEDIGENE | 0,036 | -10,84 % | MEDIGENE AG - Schwäche mit Signalcharakter | ||
| QIAGEN | 28,350 | -1,56 % | BARCLAYS stuft QIAGEN NV auf 'Equal Weight' | LONDON (dpa-AFX Analyser) - Die britische Investmentbank Barclays hat die Einstufung für Qiagen nach endgültigen Quartalszahlen auf "Equal Weight" mit einem Kursziel von 38 US-Dollar belassen. Analyst... ► Artikel lesen | |
| MODERNA | 43,070 | -5,06 % | Moderna: Hantavirus-Schlagzeilen treiben Impfstoff-Aktie an | Das Hantavirus ist auf einem kleinen Kreuzfahrtschiff im Atlantik ausgebrochen. Die Weltgesundheitsorganisation (WHO) hat inzwischen fünf von acht Verdachtsfällen als offizielle Fälle bestätigt. Aus... ► Artikel lesen | |
| PAION | 0,073 | -24,08 % | XFRA MISTRADE ANTRAG IN ISIN DE000A3E5EG5 WURDE STATTGEGEBEN | Dem Mistrade-Antrag wurde stattgegeben. Folgende Geschaefte in der ISIN DE000A3E5EG5, Wertpapier-Name: PAION AG INH O.N., wurden aufgehoben:ISIN Datum Zeit Volumen PreisDE000A3E5EG5 02.02.2026 08:13:40 300 0... ► Artikel lesen | |
| VALNEVA | 2,553 | -0,51 % | Valneva-Aktie vor Zahlen: Was ist am 13. Mai zu erwarten? | Die Aktie von Valneva bleibt auch nach der jüngsten Kapitalspritze unter Druck. Nachdem bereits in unserem vorherigen Artikel auf die begrenzten Erholungschancen hingewiesen wurde, bestätigt die aktuelle... ► Artikel lesen | |
| AMGEN | 285,05 | -0,49 % | Maria Elvira Salazar trades in Amgen, Carrier Global, and Microsoft stocks | ||
| EPIGENOMICS | 0,930 | 0,00 % | PTA-HV: Epigenomics AG: Einladung zur ordentlichen Hauptversammlung | DJ PTA-HV: Epigenomics AG: Einladung zur ordentlichen Hauptversammlung
Hauptversammlung gemäß -- 121 Abs. 4a AktG
Epigenomics AG: Einladung zur ordentlichen Hauptversammlung
Heidelberg... ► Artikel lesen | |
| NOVAVAX | 7,960 | -2,93 % | Novavax, Inc.: Novavax Reports First Quarter 2026 Financial Results and Operational Highlights | Total revenue of $140 million in the first quarter of 2026
Pfizer agreement announced in January for non-exclusive license to utilize Matrix-M® in two infectious... ► Artikel lesen | |
| STRYKER | 257,30 | +2,55 % | Stryker Corporation: Stryker declares an $0.88 per share quarterly dividend | Portage, Michigan, May 07, 2026 (GLOBE NEWSWIRE) -- Stryker (NYSE:SYK) announced that its Board of Directors has declared a quarterly dividend of $0.88 per share payable July 31, 2026, to shareholders... ► Artikel lesen | |
| BIOGEN | 170,84 | +0,57 % | Eisai, Biogen Say FDA Extends Review Of LEQEMBI IQLIK As A Starting Dose For Early AD | WESTON (dpa-AFX) - Eisai Co. Ltd (ESALY.PK) and Biogen Inc. (BIIB) on Friday said the U.S. Food and Drug Administration has extended by three months the review of LEQEMBI IQLIK as a starting... ► Artikel lesen | |
| BIOFRONTERA | 2,410 | +0,84 % | PTA-HV: Biofrontera AG: Einladung zur ordentlichen Hauptversammlung | DJ PTA-HV: Biofrontera AG: Einladung zur ordentlichen Hauptversammlung
Hauptversammlung gemäß -- 121 Abs. 4a AktG
Biofrontera AG: Einladung zur ordentlichen Hauptversammlung
Leverkusen... ► Artikel lesen | |
| HEIDELBERG PHARMA | 2,790 | +0,72 % | Mesoblast Aktie: Warum sich der Blick auf diesen Biotech-Player jetzt lohnt - und was Anleger von Merck, Regeneron & Co. lernen können |
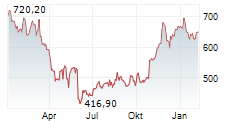
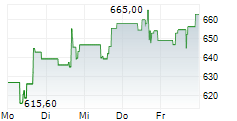
REGENERON PHARMACEUTICALS INC gehört der Branche Biotechnologie an. Die relative Kursveränderung beträgt aktuell -0,84 % bei einem Aktienkurs von 610,90 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu REGENERON PHARMACEUTICALS INC finden Sie auf Wallstreet Online.