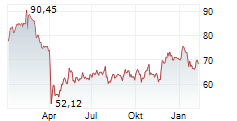
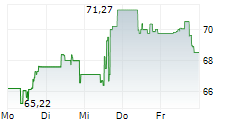
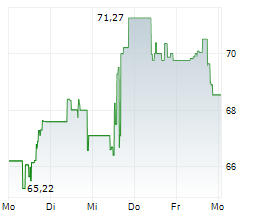
WASHINGTON (dpa-AFX) - GE HealthCare (GEHC) announced the submission of its next-generation SIGNAT MRI technology to the U.S. Food and Drug Administration (FDA) for 510(k) clearance. The new systems are designed to enhance efficiency, improve patient experience, and advance sustainability in medical imaging.
The SIGNA Bolt, a new 3T MRI scanner, features a next-generation gradient system engineered to deliver precision diagnostics, greater clinical efficiency, and advanced research capabilities. This innovation aims to support healthcare providers in achieving faster, more accurate imaging results.
The SIGNA Sprint with Freelium, a sealed magnet 1.5T MRI system, reimagines traditional MRI design by combining sustainability with exceptional image quality. With operational autonomy, it is intended to expand access to MRI services while reducing environmental impact.
Both systems are powered by SIGNA One, an AI-driven ecosystem of workflow solutions that streamline MRI exams from planning to scanning and beyond. By reducing inefficiencies and supporting clinical workflows, SIGNA One is expected to transform the MRI experience for both patients and providers.
Copyright(c) 2025 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2025 AFX News