WASHINGTON (dpa-AFX) - Medtronic plc (MDT) said Monday it has received approval from the U.S. Food and Drug Administration for an expanded indication of its OmniaSecure defibrillation lead.
The device is now approved for placement in the left bundle branch (LBB) area, enabling use in conduction system pacing (CSP), which more closely mimics the heart's natural electrical activity. The expanded indication also supports left bundle branch optimized cardiac resynchronization therapy (LOT-CRT), a newer approach combining CSP with left ventricular pacing to improve patient outcomes.
The approval was supported by data from the global LEADR LBBAP trial, which demonstrated the lead's safety and effectiveness.
The OmniaSecure lead connects to implantable cardioverter-defibrillators (ICDs) or cardiac resynchronization therapy defibrillators (CRT-Ds) to treat life-threatening ventricular tachyarrhythmias, ventricular fibrillation, and bradyarrhythmias.
Medtronic said the device is the first defibrillation lead approved for placement in the LBB area and, at 4.7 French (1.66 mm), the smallest diameter defibrillation lead available. It is also approved for use in adults and adolescent patients aged 12 and older.
The expanded indication adds to Medtronic's portfolio of FDA-approved conduction system pacing technologies, including the SelectSecure Model 3830 pacing lead, as well as the recently cleared C320LBB delivery catheter and 5944RL rotatable connector.
Following earlier approval for placement in traditional right ventricular locations, the OmniaSecure defibrillation lead was commercially launched in the U.S. in January 2026.
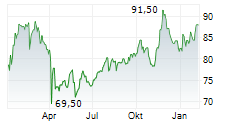
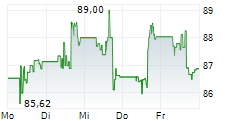
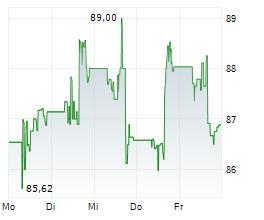
Medtronic shares closed at $86.16, down 0.82%.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News