SOUTH SAN FRANCISCO (dpa-AFX) - Roche (RO.SW, ROG.SW) announced that the cobas MPX-E assay, a qualitative in-vitro test for the detection and discrimination of Human Immunodeficiency Virus and Hepatitis C, B, and E viruses, is now available in countries accepting the CE mark. The new test delivers faster turnaround times and allows labs to implement Hepatitis E screening without requiring additional instrumentation.
The company said the assay runs on the fully automated cobas x800 systems, which are already used by laboratories worldwide to run more than 10 million tests per month.
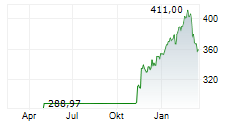
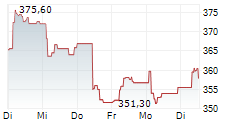
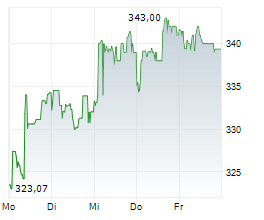
At last close, shares of Roche were trading at 312.50 Swiss francs.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News