PARIS (dpa-AFX) - Sanofi (SNY, SAN.PA) said the European Commission has granted a conditional marketing authorisation for Rezurock or belumosudil for the treatment of chronic graft-versus-host disease in adults and in children aged 12 years and older with a body weight of at least 40 kg. The conditional marketing authorisation is contingent on completion of a confirmatory, randomised, controlled study. This follows the positive opinion by the CHMP issued on 30 January 2026.
The company said the approval is based on safety and efficacy results from several clinical studies and real-world evidence. In addition to the EU, Rezurock is approved in 20 countries.
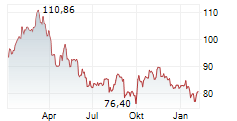
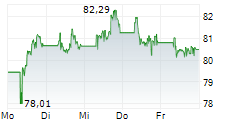
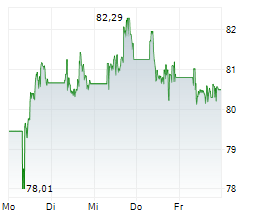
At last close, shares of Sanofi were trading at 81.90 euros, down 0.11%.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News