THOUSAND OAKS (dpa-AFX) - Amgen (AMGN) announced positive topline results from a Phase 3 trial of TEPEZZA administered by subcutaneous injection via an on-body injector in participants with moderate-to-severe active Thyroid Eye Disease. The trial met its primary endpoint in moderate-to-severe active TED, showing a statistically significant and clinically meaningful 77% proptosis response rate during the 24-week placebo-controlled period. Full results will be presented at an upcoming medical congress.
'These results extend and support the best-in-class efficacy of TEPEZZA for people living with Thyroid Eye Disease, now with subcutaneous administration delivering IV-level efficacy,' said Jay Bradner, executive vice president of Research and Development at Amgen.
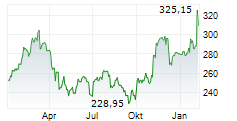
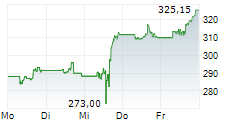
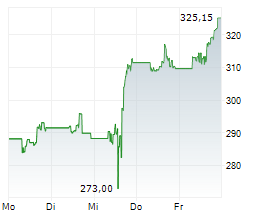
Amgen shares are currently trading at $344.11, down 1.10 percent.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News