PARIS (dpa-AFX) - Sanofi's (SNY, SNYNF,SAN.PA) protein-based COVID-19 vaccine, Nuvaxovid (NVX-CoV2705), demonstrated a significantly better tolerability profile compared to Moderna's latest mRNA vaccine, mNEXSPIKE (mRNA-1283), in the first head-to-head, double-blind, randomized phase 4 study conducted in adults.
The company said the COMPARE study found that Nuvaxovid recipients experienced fewer and less severe systemic side effects such as fatigue, headache, and fever. Severe systemic symptoms that disrupted daily activities occurred in fewer than one in ten Nuvaxovid recipients, compared to one in five among mNEXSPIKE recipients. Severe local reactions at the injection site were also rare with Nuvaxovid, occurring more than 75% less frequently than with mNEXSPIKE.
The study's primary endpoint-probability of experiencing at least one systemic reaction within seven days-was met with statistical significance. Results showed 91.6% of mNEXSPIKE recipients experienced systemic reactions versus 83.6% of Nuvaxovid recipients. Moderate-to-severe systemic symptoms affected 61.3% of mNEXSPIKE recipients compared to 43.1% of Nuvaxovid recipients, while moderate-to-severe local symptoms were reported by 58.7% of mNEXSPIKE recipients versus 38.7% of Nuvaxovid recipients.
Beyond clinical measures, patient-reported outcomes reinforced Nuvaxovid's favorable tolerability. Participants reported less disruption to work, school, recreational activities, and caregiving responsibilities following vaccination. Importantly, Nuvaxovid recipients were nearly twice as likely as those receiving mNEXSPIKE to say they would choose the same vaccine again in the future.
These findings highlight how tolerability influences vaccination behavior, with many participants scheduling their shots around anticipated side effects. By offering a more comfortable vaccination experience, Nuvaxovid may help strengthen vaccine confidence and encourage routine immunization uptake.
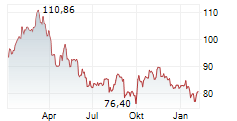
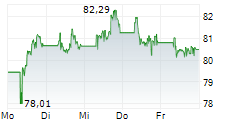
At the close of trading on April 17, Sanofi's stock price stood at $48.06, reflecting a gain of $0.34 or 0.71%. In after-hours trading, the stock continued to edge higher, reaching $48.25 , an increase of $0.19 or 0.40%.
For More Such Health News, visit rttnews.com
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News