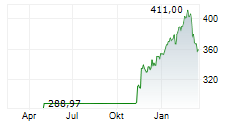
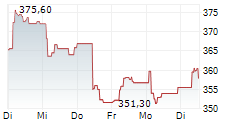
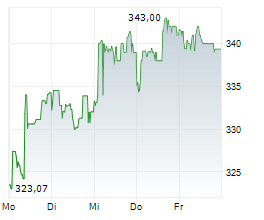
SOUTH SAN FRANCISCO (dpa-AFX) - Genentech, a unit of Roche Holding AG (RHHBY), announced Tuesday that the US Food and Drug Administration (FDA) has accepted its supplemental Biologics License Application for Gazyva for the treatment of systemic lupus erythematosus (SLE).
The filing is supported by positive results from the Phase III ALLEGORY study, which showed a statistically significant and clinically meaningful improvement in the primary endpoint of SLE Responder Index 4 at 52 weeks, a measure assessing disease severity, symptoms and physical condition.
A decision from the regulator is expected by December 2026.
Gazyva is already approved in the U.S. and EU for the treatment of adults with lupus nephritis.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News