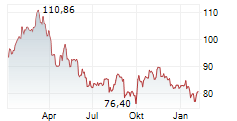
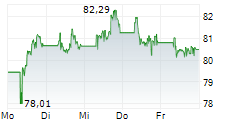
PARIS (dpa-AFX) - Sanofi SA (SNY) on Wednesday said the U.S. Food and Drug Administration has approved Tzield to expand its indication from patients aged eight years and older to those as young as one year, to delay the onset of stage 3 Type 1 Diabetes in individuals diagnosed with stage 2 T1D.
The approval is supported by one-year data from the PETITE-T1D phase 4 study.
Tzield is also under FDA review for a potential indication to delay the progression of stage 3 T1D in patients aged eight years and older who have been recently diagnosed with stage 3 disease.
The drug is approved in the European Union (under the name Teizield), the United Kingdom, China, Canada, Israel, Saudi Arabia, the UAE, Kuwait, and Brazil to delay the onset of stage 3 T1D in adults and pediatric patients aged eight years and older with stage 2 T1D. Other regulatory reviews are ongoing.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News