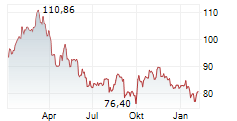
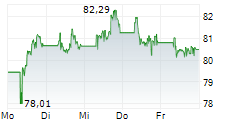
PARIS (dpa-AFX) - Sanofi SA (SNY) on Wednesday said the U.S. Food and Drug Administration has extended by up to three months the timeline to review Sarclisa subcutaneous (SC) formulation in combination with standard-of-care to treat multiple myeloma across all currently approved U.S. indications of its intravenous (IV) formulation.
The new target action date is July 23, 2026.
If approved, Sarclisa would be the first anticancer treatment administered through an on-body injector (OBI), Sanofi said.
Additionally, On March 26, 2026, the European Medicines Agency committee had recommended the approval of Sarclisa as a subcutaneous treatment for multiple myeloma. A final decision is expected in the coming months.
Sarclisa is approved in nearly 60 countries for certain patients with newly diagnosed and relapsed or refractory multiple myeloma.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News