PARIS (dpa-AFX) - The FDA has approved Dupixent for the treatment of children aged two to 11 years with chronic spontaneous urticaria who remain symptomatic despite histamine-1 antihistamine treatment. This expands the previous approval for Dupixent in adults and adolescents aged 12 years and older. The approval is based primarily on data from the LIBERTY-CUPID clinical study program.
Dupilumab is being jointly developed by Sanofi (SNY, SAN.PA) and Regeneron (REGN) under a global collaboration agreement. Dupixent is now approved for nine different allergy-related conditions, from asthma to atopic dermatitis. In addition to the currently approved indications, Sanofi and Regeneron are studying dupilumab in a broad range of diseases driven by type 2 inflammation or other allergic processes in phase 3 studies.
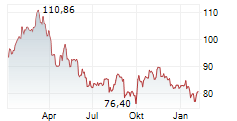
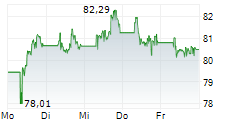
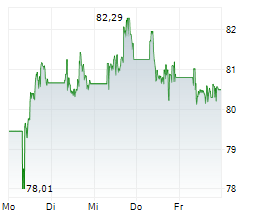
At last close, shares of Sanofi were trading at 80.78 euros, down 0.30%.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News