LONDON (dpa-AFX) - Regeneron Pharmaceuticals (REGN) announced the FDA has granted accelerated approval for Otarmeni, the first gene therapy and second new molecular entity approved under the FDA Commissioner's National Priority Voucher program. The FDA approval is based on results from the pivotal CHORD trial. Regeneron will provide Otarmeni at no cost to clinically eligible individuals in the U.S.
Otarmeni is an adeno-associated virus vector-based gene therapy indicated for the treatment of pediatric and adult patients with severe-to-profound and profound sensorineural hearing loss associated with molecularly confirmed biallelic variants in the OTOF gene, preserved outer hair cell function, and no prior cochlear implant in the same ear.
Separately, Regeneron Pharmaceuticals announced an agreement with the U.S. government to help lower prices of several of its current and future life-changing medicines and offer an important new medicine for free in the U.S. Regeneron will lower Medicaid prices based on those in other developed countries and will align U.S. prices of future medicines with prices set in that defined group of other countries. Regeneron will also utilize the TrumpRx.gov platform to allow eligible patients to directly purchase Praluent, Regeneron's PCSK9 inhibitor medicine, at the most-favored nation price. Additional terms of the agreement remain confidential.
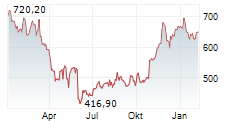
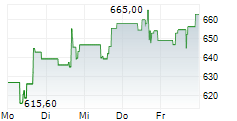
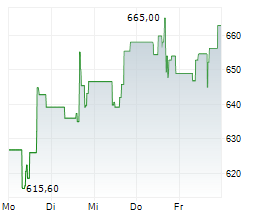
At last close, shares of Regeneron Pharmaceuticals were trading at $766.02, up 2.6%.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News