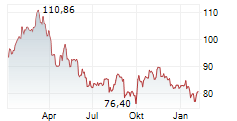
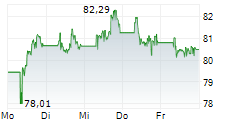
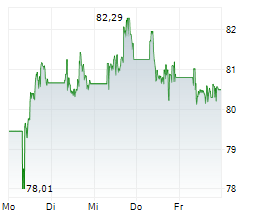
PARIS (dpa-AFX) - Sanofi SA (SNY) on Friday said the European Medicines Agency's Committee for Medicinal Products for Human Use has recommended approval of Cenrifki for treating secondary progressive multiple sclerosis (SPMS) in patients without relapses in the past two years.
A final decision from the agency is expected in the coming months.
The positive opinion is based on data from the HERCULES Phase 3 study in non-relapsing SPMS, supported by findings from the GEMINI 1 and GEMINI 2 Phase 3 studies in relapsing multiple sclerosis.
Sanofi said additional regulatory submissions for Cenrifki are currently under review in multiple countries worldwide.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News