NEW BRUNSWICK (dpa-AFX) - Johnson & Johnson (JNJ) on Tuesday announced positive results from the Phase 3 FUZION study evaluating TREMFYA in adults with active perianal fistulizing Crohn's disease.
The study met its primary endpoint of combined fistula remission at Week 24, defined as complete closure of all external fistula openings with no drainage, no new fistulas, and no evidence of underlying fluid collections on MRI.
The treatment showed statistically significant improvements compared with placebo, and adverse events through 24 weeks were consistent with the known safety profile of TREMFYA in Crohn's disease.
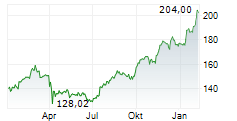
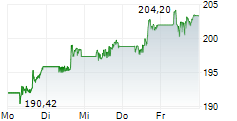
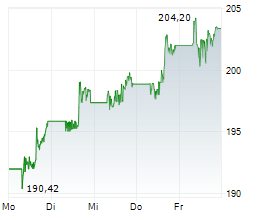
JNJ shares closed at $224.20 on Monday, down 1.32%.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News