WASHINGTON (dpa-AFX) - Regeneron Pharmaceuticals Inc. (REGN) announced results from its Phase 3 trial evaluating two dose levels of fianlimab, a LAG-3 inhibitor, in combination with cemiplimab, a PD-1 inhibitor, as a first-line treatment for patients with unresectable locally advanced or metastatic melanoma. The study did not achieve statistical significance for the primary endpoint of progression-free survival (PFS) improvement compared with pembrolizumab monotherapy.
No new safety signals were identified with the fianlimab combination. However, a numeric improvement of 5.1 months in median PFS was observed for the high-dose fianlimab combination compared to pembrolizumab.
A separate Phase 3 head-to-head trial is ongoing, evaluating the high-dose fianlimab combination against Opdualag (nivolumab and relatlimab-rmbw) in first-line unresectable or metastatic melanoma.
The randomized, double-blind Phase 3 trial is investigating the combination of fianlimab and cemiplimab versus pembrolizumab in patients 12 years of age or older with unresectable locally advanced or metastatic melanoma who have not received a previous systemic treatment for advanced disease. The trial enrolled 1,546 patients who were randomized to receive either: 1600 mg fianlimab and 350 mg cemiplimab (high-dose combination) every 3 weeks; 400 mg fianlimab and 350 mg cemiplimab (low-dose combination) every 3 weeks; placebo and 200 mg pembrolizumab every 3 weeks; or placebo and 350 mg cemiplimab every 3 weeks.
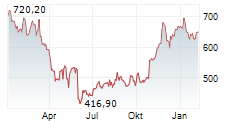
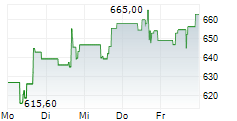
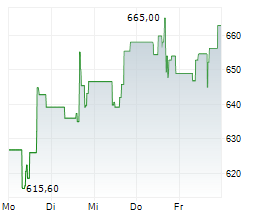
REGN closed Friday's regular trading at $698.25 down $14.62 or 2.05%.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News