| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 8,800 | 11,100 | 17:37 |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| Do | Oppenheimer initiates Adlai Nortye stock coverage with Outperform | 1 | Investing.com | ||
| 01.05. | H.C. Wainwright Sees "Potential Best-in-Class Potency" for Adlai Nortye's (ANL) Lead Asset, Lifts PT | 2 | Insider Monkey | ||
| 16.04. | Adlai Nortye Ltd. - 6-K, Report of foreign issuer | - | SEC Filings | ||
| 16.04. | Adlai Nortye jumps 19% on $150M private equity financing | 1 | Seeking Alpha | ||
| 16.04. | Adlai Nortye raises $150M in oversubscribed private placement | 1 | Investing.com | ||
| 16.04. | Adlai Nortye sichert sich 150 Mio. US-Dollar durch überzeichnete Privatplatzierung | 1 | Investing.com Deutsch | ||
| 16.04. | Adlai Nortye Ltd.: Adlai Nortye Announces $150.0 Million Private Placement Equity Financing | 3 | GlobeNewswire (USA) | ||
| ADLAI NORTYE Aktie jetzt für 0€ handeln | |||||
| 10.04. | Adlai Nortye Ltd. - 20-F, Annual and transition report of foreign private issuers | 1 | SEC Filings | ||
| 01.04. | Adlai Nortye Ltd. - 6-K, Report of foreign issuer | 3 | SEC Filings | ||
| 23.02. | XFRA AUFHEBUNG VON AMTS WEGEN IN US00704R1095, CA57778R1001, US5949728530, CA83179X1087 | 774 | Xetra Newsboard | Folgende Geschaefte in der ISIN US00704R1095, Wertpapier-Name: ADLAI NORTYE LTD. SP.ADR, wurden von Amts wegen aufgehoben:Folgende Geschaefte in der ISIN CA57778R1001, Wertpapier-Name: MAX POWER MINING... ► Artikel lesen | |
| 13.02. | H.C. Wainwright upgrades Adlai Nortye stock rating to Buy with $16 target | 1 | Investing.com | ||
| 12.02. | Adlai Nortye Ltd. - 6-K, Report of foreign issuer | 1 | SEC Filings | ||
| 12.02. | Adlai Nortye Ltd.: Adlai Nortye Announces First Patient Enrolled in Global Phase 1 Trial of Pan-RAS (ON) Inhibitor AN9025 for Solid Tumors Harboring RAS Mutations | 225 | GlobeNewswire (Europe) | SINGAPORE and NORTH BRUNSWICK, N.J. and HANGZHOU, China, Feb. 12, 2026 (GLOBE NEWSWIRE) -- Adlai Nortye Ltd. (NASDAQ: ANL) (the "Company" or "Adlai Nortye"), a clinical-stage biotechnology company... ► Artikel lesen | |
| 03.02. | Adlai Nortye Ltd. - 6-K, Report of foreign issuer | 2 | SEC Filings | ||
| 03.02. | Adlai Nortye Leaps On Stock Deal | 3 | Baystreet.ca | ||
| 03.02. | Adlai Nortye jumps 25%, raises $140M via private placement | 2 | Seeking Alpha | ||
| 03.02. | Adlai Nortye Ltd.: Adlai Nortye Announces $140.0 Million Private Placement Equity Financing | 243 | GlobeNewswire (Europe) | SINGAPORE and NORTH BRUNSWICK, N.J. and HANGZHOU, China, Feb. 03, 2026 (GLOBE NEWSWIRE) -- Adlai Nortye Ltd. (Nasdaq: ANL) (the "Company" or "Adlai Nortye"), a clinical-stage biotechnology company... ► Artikel lesen | |
| 29.01. | Lucid Capital Markets initiates coverage on Adlai Nortye stock with Buy rating | 8 | Investing.com | ||
| 29.12.25 | Adlai Nortye Stock Rises On Exclusive AN9025 China Licensing Deal With ASK Pharm | 2 | RTTNews | ||
| 29.12.25 | Adlai Nortye vergibt Lizenz für Krebsmedikament in 230-Millionen-Dollar-Deal an ASK Pharm | 13 | Investing.com Deutsch |
1 2 Weiter >>
23 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 76,05 | -0,33 % | Biontech: Doppelte Hochstufung der Aktie | Die Analysten der UBS schauen sich die Onkologie-Pipeline von Biontech an. Was sie dort sehen, hinterlässt offenbar einen sehr positiven Eindruck bei den Experten. Noch sind viele Anleger hinsichtlich... ► Artikel lesen | |
| OCUGEN | 1,130 | -2,42 % | Ocugen's Gene-Agnostic Eye Therapies Could Reshape Late-Stage Care | ||
| VIKING THERAPEUTICS | 25,450 | +0,99 % | Bull vs. Bear: The Quick Take on Viking Therapeutics Stock | ||
| INFLARX | 1,853 | -1,12 % | ANALYSE: InflaRx NV: Könnte die Aktie jetzt komplett einbrechen? - Was Sie jetzt noch wissen müssen | ||
| EDITAS MEDICINE | 2,480 | +2,48 % | Editas Medicine, Inc.: Editas Medicine Reports New Preclinical Data Demonstrating Progress of EDIT-401 as Potential Treatment for Hyperlipidemia at the American Society of Gene and Cell Therapy 2026 Annual Meeting | CAMBRIDGE, Mass., May 14, 2026 (GLOBE NEWSWIRE) -- Editas Medicine, Inc. (Nasdaq: EDIT), a pioneering gene editing company developing transformative medicines for serious diseases, shared new preclinical... ► Artikel lesen | |
| SAREPTA THERAPEUTICS | 14,140 | +1,14 % | Biotech-Player: Zelltherapie als Comeback-Story im Milliardenmarkt. Neue Ideen nach Corona von BioNTech, Qiagen und Biotech AG | ||
| CARDIOL THERAPEUTICS | 0,983 | -1,01 % | Original-Research: Cardiol Therapeutics Inc (von First Berlin Equity Research GmbH): BUY | Original-Research: Cardiol Therapeutics Inc - from First Berlin Equity Research GmbH
30.04.2026 / 13:07 CET/CEST
Dissemination of a Research, transmitted by EQS News - a service of EQS Group.
The... ► Artikel lesen | |
| BASILEA | 54,40 | +0,37 % | Basilea Pharmaceutica International Ltd, Allschwil: Basilea erhält weitere USD 13 Mio. von BARDA zur Entwicklung des neuartigen Antibiotikums Ceftibuten-Ledaborbactam | Ad hoc-Mitteilung gemäss Art. 53 KR
Allschwil, 28. Mai 2026
Basilea Pharmaceutica AG, Allschwil (SIX: BSLN), ein biopharmazeutisches Unternehmen mit bereits vermarkteten Produkten und dem Ziel... ► Artikel lesen | |
| ARROWHEAD PHARMACEUTICALS | 63,00 | +3,45 % | Arrowhead Pharmaceuticals, Inc.: Arrowhead Pharmaceuticals Presents New Positive Clinical Cardiometabolic Data at the 94th European Atherosclerosis Society (EAS) Congress | New data support potential use of plozasiran without dose adjustment in patients with moderate-to-severe renal or moderate hepatic impairment
Case report suggests that preconception exposure to... ► Artikel lesen | |
| TELIX PHARMACEUTICALS | 7,559 | -1,18 % | Telix Pharmaceuticals Limited: Telix and United Imaging Announce Strategic Theranostics Collaboration | Partnership to evaluate Telix's innovative precision medicine products and candidates in combination with United Imaging's advanced scanner platforms.Initial focus on TLX101-Px (Pixclara®1, Floretyrosine... ► Artikel lesen | |
| ONCO-INNOVATIONS | 0,560 | -2,78 % | Onco-Innovations Limited: Onco-Innovations Advances 1kg Scale-Up of B4 Precursor Supporting ONC010 Manufacturing and Clinical Development Program | Also Announces Marketing Program VANCOUVER, BC / ACCESS Newswire / June 2, 2026 / Onco-Innovations Limited (CBOE CA:ONCO)(OTCQB:ONNVF)(Frankfurt:W1H) (WKN: A3EKSZ) ("Onco" or the "Company") announces... ► Artikel lesen | |
| TWIST BIOSCIENCE | 61,28 | +1,52 % | LenioBio GmbH: LenioBio and Twist Bioscience Enter into a Collaboration to Further Enable AI drug-discovery | LenioBio GmbH, a TechBio company commercializing its ALiCE cell-free protein expression platform, today announced a collaborative agreement with Twist Bioscience Corporation. The collaboration will... ► Artikel lesen | |
| REPLIGEN | 102,60 | -1,20 % | Wolfe Research initiates Repligen stock coverage with outperform rating | ||
| UNIQURE | 25,250 | +9,78 % | uniQure-Aktie springt um 20 % nach positiven FDA-Nachrichten von Wettbewerber Replimune | ||
| REVOLUTION MEDICINES | 133,15 | +2,30 % | Neue Krebs-Hoffnung: Revolution Medicines zieht 20% an: Was hinter dem Krebsdurchbruch steckt | © Foto: Frank Rumpenhorst - dpaEin Biotech-Wirkstoff schafft, woran die Onkologie jahrzehntelang gescheitert ist: Pankreas-Patienten überleben im Median mehr als ein Jahr. Die Aktie von Revolution Medecines... ► Artikel lesen |
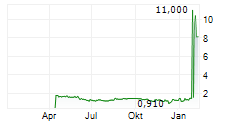
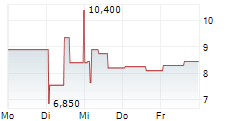
Das Unternehmen ADLAI NORTYE LTD ADR kann der Branche Biotechnologie zugeordnet werden. Mit einer aktuellen Kursveränderung von -0,400 (-4,19 %) liegt der Kurs bei 9,150 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu ADLAI NORTYE LTD ADR finden Sie auf Wallstreet Online.