- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 14.05. | Fortress Biotech, Inc. - 10-Q, Quarterly Report | 1 | SEC Filings | ||
| 14.05. | Fortress Biotech GAAP EPS of $2.82, revenue of $16.04M misses by $0.3M | 3 | Seeking Alpha | ||
| 14.05. | Fortress Biotech, Inc. - 8-K, Current Report | 1 | SEC Filings | ||
| 14.05. | Fortress Biotech, Inc.: Fortress Biotech Reports First Quarter 2026 Financial Results and Recent Corporate Highlights | 619 | GlobeNewswire (Europe) | ZYCUBO approved by FDA to treat Menkes disease in the United States; Fortress subsidiary Cyprium Therapeutics closed the sale of its Rare Pediatric Disease Priority Review Voucher (PRV) for $205 million... ► Artikel lesen | |
| 31.03. | Fortress Biotech GAAP EPS of -$0.07 misses by $0.39, revenue of $63.26M misses by $10.08M | 6 | Seeking Alpha | ||
| 31.03. | Fortress Biotech, Inc.: Fortress Biotech Reports 2025 Financial Results and Recent Corporate Highlights | 739 | GlobeNewswire (Europe) | ZYCUBOapproved by FDA to treat Menkes disease in the United States; Fortress subsidiary Cyprium Therapeutics sold its Rare Pediatric Disease Priority Review Voucher (PRV) for gross proceeds of $205... ► Artikel lesen | |
| 31.03. | Fortress Biotech, Inc. - 10-K, Annual Report | - | SEC Filings | ||
| 30.03. | Fortress Biotech, Inc.: Fortress Biotech's Subsidiary Cyprium Therapeutics Closes Sale of Rare Pediatric Disease Priority Review Voucher for $205 Million | 363 | GlobeNewswire (Europe) | MIAMI, March 30, 2026 (GLOBE NEWSWIRE) -- Fortress Biotech, Inc. (Nasdaq: FBIO) ("Fortress") and its majority-owned subsidiary, Cyprium Therapeutics, Inc. ("Cyprium"), today announced the closing... ► Artikel lesen | |
| FORTRESS BIOTECH INC PREF Aktie jetzt für 0€ handeln | |||||
| 23.02. | Fortress Biotech, Inc. - 8-K, Current Report | 5 | SEC Filings | ||
| 23.02. | Fortress Biotech, Inc.: Fortress Biotech's Subsidiary Cyprium Therapeutics Enters into Agreement to Sell Rare Pediatric Disease Priority Review Voucher for $205 Million | 472 | GlobeNewswire (Europe) | MIAMI, Feb. 23, 2026 (GLOBE NEWSWIRE) -- Fortress Biotech, Inc. (Nasdaq: FBIO) ("Fortress") and its majority-owned subsidiary, Cyprium Therapeutics, Inc. ("Cyprium"), today announced that Cyprium... ► Artikel lesen | |
| 16.01. | Weekly Buzz: Lyra Therapeutics Slashes Jobs; FDA Okays Fortress Biotech's ZYCUBO; Boston Scientific To Acquire Penumbra; BriaCell Therapeutics On Watch | 830 | AFX News | NEW BRUNSWICK (dpa-AFX) - This week, the biotech sector saw a steady stream of high-impact news, including a major workforce reduction and a fresh round of regulatory clearances spanning diabetes... ► Artikel lesen | |
| 14.01. | Fortress Biotech receives FDA approval for Zycubo to treat Menkes disease | 13 | Pharmaceutical Technology | ||
| 13.01. | Fortress Bio gains on FDA approval of copper replacement therapy | 6 | Seeking Alpha | ||
| 13.01. | Fortress Biotech, Inc. - 8-K, Current Report | 2 | SEC Filings | ||
| 13.01. | Fortress Biotech, Inc.: Fortress Biotech and Cyprium Therapeutics Announce U.S. FDA Approval of ZYCUBO (copper histidinate), the First and Only Approved Treatment for Menkes Disease in the United States | 883 | GlobeNewswire (Europe) | Rare Pediatric Disease Priority Review Voucher (PRV) granted by FDA at approval to be transferred from Sentynl Therapeutics to Cyprium Cyprium eligible to receive tiered royalties and up to $129 million... ► Artikel lesen | |
| 15.12.25 | FDA sets new PDUFA date for Fortress Biotech's Menkes disease therapy | 31 | Investing.com | ||
| 15.12.25 | Fortress Biotech, Inc. - 8-K, Current Report | 3 | SEC Filings | ||
| 14.11.25 | Fortress Biotech, Inc.: Fortress Biotech Reports Third Quarter 2025 Financial Results and Recent Corporate Highlights | 782 | GlobeNewswire (Europe) | Total net revenue increased 20.5% to $17.6 million for third quarter of 2025 compared to the third quarter of 2024 Fortress subsidiary Checkpoint Therapeutics acquired by Sun Pharma; Fortress received... ► Artikel lesen | |
| 21.10.25 | Fortress Biotech, Inc.: Fortress Biotech and Subsidiary Urica Therapeutics Announce First Patients Dosed in Crystalys Therapeutics' Global Phase 3 Trials of Dotinurad for the Treatment of Gout | 352 | GlobeNewswire (Europe) | MIAMI, Oct. 21, 2025 (GLOBE NEWSWIRE) -- Urica Therapeutics, Inc. ("Urica" or the "Company"), a Fortress Biotech, Inc. (Nasdaq: FBIO) ("Fortress") subsidiary, today announced that Crystalys Therapeutics... ► Artikel lesen | |
| 01.10.25 | Fortress Biotech, Inc.: Fortress Biotech and Subsidiary Urica Therapeutics Announce Crystalys Therapeutics' $205 Million Series A Financing | 212 | GlobeNewswire (Europe) | Dotinurad is a next-generation URAT1 inhibitor in two Phase 3 clinical trials with potential for best-in-class safety and efficacy Urica sold dotinurad to Crystalys Therapeutics in 2024 in exchange... ► Artikel lesen |
1 2 Weiter >>
22 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| STRYKER | 265,60 | +0,15 % | Leerink cuts Stryker stock price target to $407 on model update | ||
| REGENERON PHARMACEUTICALS | 551,40 | +0,04 % | CytomX Therapeutics Inc.: CytomX Announces Expansion of Strategic Research Collaboration with Regeneron in the Field of Conditional Bispecific Therapeutics for the Treatment of Cancer | - Expanded collaboration builds upon research momentum in developing next-generation bispecific immunotherapies using CytomX's PROBODY- and Regeneron's Veloci-Bi- platforms - - CytomX to receive... ► Artikel lesen | |
| VIKING THERAPEUTICS | 24,650 | -0,20 % | XETR NEW INSTRUMENTS AVAILABLE ON XETRA - 04.06.2026 | Folgende Wertpapiere werden in den Handel an folgenden Handelsplätzen (MIC Code) im Quotation Board an der Frankfurter Wertpapierbörse (FWB) aufgenommen. Die Notierung an anderen Handelsplätzen im Quotation... ► Artikel lesen | |
| INTELLIA THERAPEUTICS | 11,240 | -4,26 % | Intellia Therapeutics, Inc. (NTLA) Buy Rating Reiterated Despite Share Offering Dilution | ||
| TEMPUS AI | 40,195 | -0,22 % | Tempus AI gründet Open-Source-Konsortium für digitale Pathologie | ||
| NUREXONE BIOLOGIC | 0,374 | 0,00 % | NurExone stärkt Patentschutz für ExoPTEN in Südkorea | ||
| PACIFIC BIOSCIENCES OF CALIFORNIA | 1,235 | -1,56 % | PACIFIC BIOSCIENCES OF CALIFORNIA, INC. - 8-K, Current Report | ||
| IMMUNITYBIO | 6,070 | +1,17 % | ImmunityBio Inc. (IBRX): One of the Most Shorted Mid-Cap Stocks to Buy Now | ||
| RECURSION PHARMACEUTICALS | 3,315 | 0,00 % | Recursion Pharmaceuticals: Recursion Reports First Quarter Financial Results and Provides Business Update | Multiple milestones achieved or on track across wholly owned and partnered programs REC-1245 (RBM39 degrader): Early clinical data in solid tumors demonstrate a well-tolerated safety profile and predictable... ► Artikel lesen | |
| GINKGO BIOWORKS | 6,904 | -2,90 % | Ginkgo Bioworks Reports First Quarter 2026 Financial Results, Completes Divestiture of Biosecurity and Continues to Scale Autonomous Lab | Ginkgo provides an update on its first quarter financial results following the divestiture of its Biosecurity business
BOSTON, May 7, 2026 /PRNewswire/ -- Ginkgo... ► Artikel lesen | |
| IOVANCE BIOTHERAPEUTICS | 3,680 | +0,41 % | Iovance Biotherapeutics, Inc.: Iovance's Amtagvi (lifileucel) Granted Approval for the Treatment of Advanced Melanoma in Australia | First T cell therapy for a solid tumor cancer and first treatment option approved in Australia for advanced melanoma after anti-PD-1 and targeted therapy SAN CARLOS, Calif., June 03, 2026 (GLOBE... ► Artikel lesen | |
| CYTOMX THERAPEUTICS | 2,586 | -0,15 % | REGN Expands Cancer Deal With CytomX for Bispecific Therapeutics | ||
| SANA BIOTECHNOLOGY | 2,675 | -0,19 % | Sana Biotechnology, Inc. - 8-K, Current Report | ||
| BIOMEA FUSION | 1,270 | 0,00 % | Biomea Fusion, Inc.: Biomea Fusion Presents New Clinical and Translational Data for Icovamenib at the American Diabetes Association ("ADA") 86th Scientific Sessions and Announces Expansion of Ongoing Phase I BMF-650 Study | ||
| PRAXIS PRECISION MEDICINES | 254,00 | 0,00 % | Praxis Precision Medicines, Inc.: Praxis Precision Medicines Provides Vormatrigine Program Update | POWER1 Study in highly refractory patients with focal onset seizures did not meet its primary success measure Secondary measure, the 50% response rate, was met and seizure reduction during the second... ► Artikel lesen |
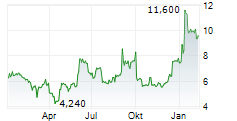
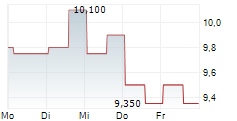
Das Unternehmen FORTRESS BIOTECH INC PREF kann der Branche Biotechnologie zugeordnet werden. Die relative Kursveränderung beträgt aktuell +6,59 % bei einem Aktienkurs von 17,800 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu FORTRESS BIOTECH INC PREF finden Sie auf Wallstreet Online.