| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
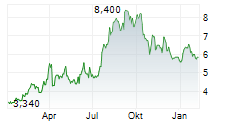
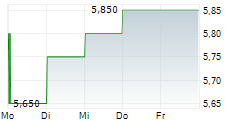
| 6,850 | 7,000 | 13:05 | |
| 6,750 | 7,100 | 22.05. |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 29.04. | KEYMED BIO-B (02162): FORM OF PROXY FOR USE AT ANNUAL GENERAL MEETING | - | HKEx | ||
| 29.04. | KEYMED BIO-B (02162): NOTICE OF ANNUAL GENERAL MEETING | - | HKEx | ||
| KEYMED BIOSCIENCES Aktie jetzt für 0€ handeln | |||||
| 29.04. | KEYMED BIO-B (02162): (1) GENERAL MANDATES TO ISSUE SHARES AND REPURCHASE SHARES; (2) RE-ELECTION OF DIRECTORS; (3) RE-APPOINTMENT OF AUDITOR; AND (4) ... | 1 | HKEx | ||
| 29.04. | KEYMED BIO-B (02162): 2025 ANNUAL REPORT | 1 | HKEx | ||
| 15.04. | KEYMED BIO-B (02162): SUPPLEMENTAL ANNOUNCEMENT TO DISCLOSEABLE TRANSACTION DISPOSAL OF EQUITY INTEREST IN OURO MEDICINES | - | HKEx | ||
| 02.04. | A Healthy Return: Drug Spin-off Nets Keymed Up To $320M | 2 | Benzinga.com | ||
| 26.03. | Keymed Biosciences Announces 2025 Annual Results and Business Updates | 301 | PR Newswire | BEIJING, March 26, 2026 /PRNewswire/ -- Keymed Biosciences (HKEX: 02162) announced its 2025 annual results.
Total revenue in 2025 was approximately RMB 720 million, a 67% year-on-year... ► Artikel lesen | |
| 26.03. | KEYMED BIO-B (02162): ANNUAL RESULTS ANNOUNCEMENT FOR THE YEAR ENDED DECEMBER 31, 2025 | 1 | HKEx | ||
| 23.03. | KEYMED BIO-B (02162): DISCLOSEABLE TRANSACTION DISPOSAL OF EQUITY INTEREST IN OURO MEDICINES | 1 | HKEx | ||
| 16.03. | KEYMED BIO-B (02162): DATE OF BOARD MEETING | - | HKEx | ||
| 10.03. | KEYMED BIO-B (02162): VOLUNTARY ANNOUNCEMENT - UPDATE ON COLLABORATION WITH ASTRAZENECA ON CORE PRODUCT CMG901 (AZD0901) | - | HKEx | ||
| 27.08.25 | Keymed Biosciences (02162.HK) Reports Strong Interim 2025 Results with Accelerated Commercialization and Robust R&D Momentum | 335 | PR Newswire | CHENGDU, China, Aug. 27, 2025 /PRNewswire/ -- Keymed Biosciences Inc. ("Keymed", 02162.HK) announced impressive interim results for 2025, marked by accelerated commercialization and R&D progress.
Financially... ► Artikel lesen | |
| 13.06.25 | Keymed Biosciences Announces the Latest Clinical Trial Results of CM336 Published in the New England Journal of Medicine | 897 | PR Newswire | CHENGDU, China, June 13, 2025 /PRNewswire/ -- On June 12, Keymed Biosciences Inc. (HKEX: 02162) announced that Prof. Jun Shi's research team from the Institute of Hematology and Blood Diseases... ► Artikel lesen |
13 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 79,35 | +1,67 % | BioNTech-Aktie: Gap-Close und bereit für +24%? | Die BioNTech-Aktie hat den zuletzt markanten Unterstützungsbereich um 76,40 € angelaufen und eine Reaktion gezeigt. Heute mit Up-Gap gestartet, könnte sich eine zeitnahe Schließung dieser Eröffnungslücke... ► Artikel lesen | |
| OCUGEN | 1,176 | +2,08 % | Ocugen Provides Business Update with First Quarter 2026 Financial Results | Conference Call and Webcast Today at 8:30 a.m. ET Positive 12-month data from the OCU410 Phase 2 ArMaDa clinical trial for geographic atrophy (GA) indicates a statistically significant (p About 20%... ► Artikel lesen | |
| VIKING THERAPEUTICS | 26,350 | -1,13 % | Could Viking Therapeutics Be the Next Eli Lilly? | ||
| INFLARX | 2,250 | -0,18 % | EILMELDUNG bei InflaRx NV: Was Insider zum Wochenstart bereits wissen - Was Sie vor Montag noch wissen müssen | ||
| EDITAS MEDICINE | 2,310 | -2,94 % | Editas Medicine, Inc.: Editas Medicine Reports New Preclinical Data Demonstrating Progress of EDIT-401 as Potential Treatment for Hyperlipidemia at the American Society of Gene and Cell Therapy 2026 Annual Meeting | CAMBRIDGE, Mass., May 14, 2026 (GLOBE NEWSWIRE) -- Editas Medicine, Inc. (Nasdaq: EDIT), a pioneering gene editing company developing transformative medicines for serious diseases, shared new preclinical... ► Artikel lesen | |
| SAREPTA THERAPEUTICS | 14,620 | +0,97 % | Biotech-Player: Zelltherapie als Comeback-Story im Milliardenmarkt. Neue Ideen nach Corona von BioNTech, Qiagen und Biotech AG | ||
| CARDIOL THERAPEUTICS | 1,154 | +1,05 % | Original-Research: Cardiol Therapeutics Inc (von First Berlin Equity Research GmbH): BUY | Original-Research: Cardiol Therapeutics Inc - from First Berlin Equity Research GmbH
30.04.2026 / 13:07 CET/CEST
Dissemination of a Research, transmitted by EQS News - a service of EQS Group.
The... ► Artikel lesen | |
| BASILEA | 58,60 | +0,69 % | Basilea Pharmaceutica International Ltd, Allschwil: Basilea shareholders approve all proposals of the board of directors at the 2026 annual general meeting | Allschwil, Switzerland, April 15, 2026
Basilea Pharmaceutica Ltd, Allschwil (SIX: BSLN), a commercial-stage biopharmaceutical company committed to meeting the needs of patients with severe bacterial... ► Artikel lesen | |
| ARROWHEAD PHARMACEUTICALS | 65,10 | +0,84 % | Arrowhead Pharmaceuticals, Inc.: Arrowhead Pharmaceuticals to Participate in Upcoming May 2026 Events | Arrowhead Pharmaceuticals, Inc. (NASDAQ: ARWR) today announced that it is scheduled to participate in the following upcoming events:
BofA Securities 2026 Healthcare Conference May 12-14, 2026
Type:... ► Artikel lesen | |
| TELIX PHARMACEUTICALS | 8,189 | +0,34 % | Telix Pharmaceuticals Limited: IPAX-2 Study of TLX101-Tx in First-line Glioblastoma Completes Enrolment and Confirms Dosing | IPAX-2 study of TLX101-Tx (¹³¹I-iodofalan) has completed patient enrolment.Maximum dose reached with no dose-limiting toxicities observed.TLX101-Tx is also the subject of a pivotal trial, IPAX BrIGHT... ► Artikel lesen | |
| ONCO-INNOVATIONS | 0,663 | +0,45 % | Onco-Innovations stärkt Qualitätstests für den PNKP-Inhibitor-Arzneimittelkandidaten | Vancouver, Kanada - 15. Mai 2026 / IRW-Press / Onco-Innovations Limited (CBOE CA: ONCO) (OTCQB: ONNVF) (FWB: W1H, WKN: A3EKSZ) ("Onco" oder das "Unternehmen") freut sich bekannt zugeben, dass es... ► Artikel lesen | |
| TWIST BIOSCIENCE | 52,68 | +0,96 % | Leerink raises Twist Bioscience stock price target on AI growth outlook | ||
| REPLIGEN | 93,00 | -6,21 % | Repligen Corporation: Repligen Announces Publication of the Company's 2025 Corporate Sustainability Report | ||
| UNIQURE | 20,850 | 0,00 % | uniQure Inc.: uniQure Announces First Quarter 2026 Financial Results and Provides Recent Company Updates | ~ Advancing FDA interactions on AMT-130 for Huntington's disease; Type B meeting scheduled for the second quarter of 2026 ~
~ Progressing AMT-130 toward expected UK regulatory submission; MAA on... ► Artikel lesen | |
| PHARMAMAR | 100,60 | +0,20 % | PHARMA MAR, S.A.: The Company announces the call of the Ordinary General Shareholders' Meeting and submits proposed resolutions. |
Das Unternehmen KEYMED BIOSCIENCES INC kann der Branche Biotechnologie zugeordnet werden. Der aktuelle Kurs liegt bei 6,750 Euro und damit -1,46 % im Minus.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu KEYMED BIOSCIENCES INC finden Sie auf Wallstreet Online.