| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 1,286 | 1,422 | 03.06. |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| MOLECURE Aktie jetzt für 0€ handeln |
0 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| CO.DON | 0,018 | -20,00 % | EQS-AFR: CO.DON AG i.l.: Vorabbekanntmachung über die Veröffentlichung von Finanzberichten gemäß §§ 114, 115, 117 WpHG | EQS Vorabbekanntmachung Finanzberichte: CO.DON AG i.l.
/ Vorabbekanntmachung über die Veröffentlichung von Rechnungslegungsberichten
CO.DON AG i.l.: Vorabbekanntmachung über die... ► Artikel lesen | |
| SANGAMO THERAPEUTICS | 0,126 | -100,00 % | Sangamo Therapeutics, Inc.: Sangamo Therapeutics Reports Recent Business Highlights and First Quarter 2026 Financial Results | Rolling submission of Biologics License Agreement (BLA) to U.S. Food and Drug Administration (FDA) seeking ST-920 approval is in progress. Six sites activated in Phase 1/2 STAND study evaluating ST-503... ► Artikel lesen | |
| IMMATICS | 8,685 | +0,52 % | Immatics Presents Clinical Activity and Response Dynamics of Anzu-cel PRAME Cell Therapy at 2026 ASCO Annual Meeting | One-time infusion of anzu-cel (anzutresgene autoleucel, IMA203) PRAME cell therapy induced rapid, deep and durable systemic anti-tumor activity in metastatic PD-1 relapsed cutaneous melanoma and metastatic... ► Artikel lesen | |
| ALDEYRA | 1,590 | +11,97 % | Aldeyra Therapeutics: Aufsichtsratsmitglied Martin J. Joyce tritt nicht zur Wiederwahl an | ||
| MEDIFAST | 9,950 | -1,68 % | Medifast: Hoher Cash-Bestand je Aktie trifft auf massiven Bewertungsabschlag - Einstiegschance oder Value-Falle? | Medifast verfügt über rund 15 US?Dollar Nettocash je Aktie und wird dennoch deutlich unter früheren Bewertungsniveaus gehandelt. Die Kombination aus hoher Liquidität, Dividendenrendite und potenziell... ► Artikel lesen | |
| PHARMING | 1,065 | -0,47 % | Pharming Group N.V.: Pharming Group to participate in Fireside Chat at 2026 Jefferies Global Healthcare Conference in New York | Leiden, the Netherlands, June 2, 2026: Pharming Group ("Pharming") (Euronext Amsterdam: PHARM/Nasdaq: PHAR) today announced its participation in the 2026 Jefferies Global Healthcare Conference in New... ► Artikel lesen | |
| COHERUS ONCOLOGY | 1,300 | -0,84 % | Aktionäre von Coherus Oncology stimmen für Direktorenwahl und Änderungen am Eigenkapital-Plan | ||
| ORUKA THERAPEUTICS | 50,000 | +5,04 % | H.C. Wainwright reiterates Oruka Therapeutics stock rating at buy | ||
| INNOVIVA | 18,500 | +0,54 % | Wechsel im Verwaltungsrat von Innoviva: Josephine Linden wird neues Mitglied | ||
| ARMATA PHARMACEUTICALS | 6,550 | -2,96 % | Armata Pharmaceuticals, Inc.: Armata Pharmaceuticals Announces First Quarter 2026 Results and Provides Corporate Update | Entered into secured credit agreement with Innoviva for $25 million maturing in 2029
LOS ANGELES, May 13, 2026 /PRNewswire/ -- Armata Pharmaceuticals, Inc. (NYSE... ► Artikel lesen | |
| LIQUIDIA | 47,130 | -0,55 % | Liquidia is the best performing healthcare stock in the past month | ||
| SYNDAX PHARMACEUTICALS | 15,990 | +1,23 % | H.C. Wainwright reiterates Syndax Pharmaceuticals stock rating on ASCO data | ||
| DBV TECHNOLOGIES | 3,004 | -0,46 % | DBV Technologies S.A.: Information Regarding the Total Number of Voting Rights and Total Number of Shares of the Company as of May 31, 2026 | Information Regarding the Total Number of Voting Rights and Total Number of Shares of the Company as of May 31, 2026
(Article 223-16 of the General Regulations of the Autorité des Marchés Financiers)
Market... ► Artikel lesen | |
| BRAINSTORM CELL THERAPEUTICS | 0,673 | -100,00 % | BrainStorm Cell Therapeutics Inc.: BrainStorm Cell Therapeutics Appoints Former FDA Associate Commissioner Peter J. Pitts to Board of Directors | Former Senior FDA Official Joins Board as Company Prepares to Initiate Historic Phase 3b ALS Trial Under Special Protocol Assessment NEW YORK, May 28, 2026 /PRNewswire/ -- BrainStorm Cell... ► Artikel lesen | |
| SIGA TECHNOLOGIES | 4,275 | -1,04 % | Dividendenbekanntmachungen (07.04.2026) | Unternehmen ISIN-Code Dividende (Währung) Dividende (EUR) ARCA CONTINENTAL SAB DE CV MX01AC100006 4,28 MXN 0,2085 EUR BANK OF NOVA SCOTIA CA0641491075 1,1 CAD 0,6851 EUR CAIXABANK SA ES0140609019 - 0... ► Artikel lesen |
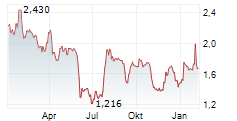
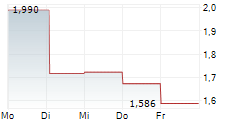
Das Unternehmen MOLECURE SA kann der Branche Biotechnologie zugeordnet werden. Der aktuelle Kurs liegt bei 1,340 Euro und damit +0,30 % im Plus.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu MOLECURE SA finden Sie auf Wallstreet Online.