| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 0,097 | 0,155 | 01.06. | |
| 0,150 | 0,158 | 04.05. |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| SANGAMO THERAPEUTICS Aktie jetzt für 0€ handeln | |||||
| 15.05. | Sangamo anticipates completing ST-920 BLA submission as early as this summer while appealing NASDAQ delisting in June | 49 | Seeking Alpha | ||
| 14.05. | Full Transcript: Sangamo Therapeutics Q1 2026 Earnings Call | 15 | Benzinga.com | ||
| 14.05. | Sangamo Therapeutics: EPS verfehlt Schätzungen um 0,02 $ - Umsatz schlechter als erwartet | 10 | Investing.com Deutsch | ||
| 14.05. | Sangamo Therapeutics (SGMO) Reports Q1 Loss, Misses Revenue Estimates | 5 | Zacks | ||
| 14.05. | Sangamo Therapeutics, Inc.: Sangamo Therapeutics Reports Recent Business Highlights and First Quarter 2026 Financial Results | 275 | GlobeNewswire (Europe) | Rolling submission of Biologics License Agreement (BLA) to U.S. Food and Drug Administration (FDA) seeking ST-920 approval is in progress. Six sites activated in Phase 1/2 STAND study evaluating ST-503... ► Artikel lesen | |
| 14.05. | SANGAMO THERAPEUTICS, INC - 8-K, Current Report | 2 | SEC Filings | ||
| 12.05. | XFRA DELETION OF INSTRUMENTS FROM BOERSE FRANKFURT - 12.05.2026 | 685 | Xetra Newsboard | The following instruments on Boerse Frankfurt do have their last trading day on 12.05.2026Die folgenden Instrumente in Boerse Frankfurt haben ihren letzten Handelstag am 12.05.2026ISIN NameAU0000182628 WINSOME... ► Artikel lesen | |
| 11.05. | XFRA GBY: AUSSETZUNG/SUSPENSION | 446 | Xetra Newsboard | DAS/ DIE FOLGENDE(N) INSTRUMENT(E) IST/ SIND AB SOFORT AUSGESETZT:THE FOLLOWING INSTRUMENT(S) IS/ ARE SUSPENDED WITH IMMEDIATE EFFECT:INSTRUMENT NAME KUERZEL/SHORTCODE ISIN BIS/UNTILSANGAMO THERAP.INC.DL-... ► Artikel lesen | |
| 30.04. | Sangamo Therapeutics, Inc.: Sangamo Therapeutics to Present Pipeline and Technology Advances at the 29th Annual Meeting of The American Society of Gene & Cell Therapy (ASGCT) | 53 | GlobeNewswire (USA) | ||
| 30.04. | SANGAMO THERAPEUTICS, INC - 10-K/A, Annual Report | 20 | SEC Filings | ||
| 29.04. | Sangamo Therapeutics, Inc.: Sangamo Therapeutics Announces Transition to Trading on OTCQB Venture Market | 384 | GlobeNewswire (Europe) | RICHMOND, Calif., April 29, 2026 (GLOBE NEWSWIRE) -- Sangamo Therapeutics, Inc. (Nasdaq: SGMO), a genomic medicine company, today announced that its common stock will transition from trading on The... ► Artikel lesen | |
| 29.04. | SANGAMO THERAPEUTICS, INC - 8-K, Current Report | 15 | SEC Filings | ||
| 31.03. | Sangamo targets summer 2026 for Fabry BLA submission while advancing neurology pipeline and partner talks | 65 | Seeking Alpha | ||
| 30.03. | SANGAMO THERAPEUTICS, INC - 8-K, Current Report | 11 | SEC Filings | ||
| 30.03. | Sangamo Therapeutics GAAP EPS of -$0.11 misses by $0.12, revenue of $14.23M misses by $26.02M | 24 | Seeking Alpha | ||
| 30.03. | Sangamo Therapeutics, Inc.: Sangamo Therapeutics Reports Recent Business Highlights And Fourth Quarter And Full Year 2025 Financial Results | 699 | GlobeNewswire (Europe) | Announced in June, positive topline results from registrational STAAR study in Fabry disease, including positive mean annualized estimated glomerular filtration rate (eGFR) slope at 52-weeks across... ► Artikel lesen | |
| 30.03. | Sangamo Therapeutics Earnings Are Imminent; These Most Accurate Analysts Revise Forecasts Ahead Of Earnings Call | 37 | Benzinga.com | ||
| 09.03. | Sangamo Therapeutics, Inc.: Sangamo Therapeutics Advances Rolling Submission of BLA to U.S. FDA for ST-920 in Fabry Disease | 1.429 | GlobeNewswire (Europe) | Data support potential of isaralgagene civaparvovec as a one-time, well tolerated and durable Fabry disease gene therapy to provide meaningful, multi-organ clinical benefits that could fundamentally... ► Artikel lesen | |
| 06.02. | Weekly Buzz: aTyr Pharma To Meet With FDA, Sangamo Therapeutics Aces Fabry Study, Aquestive Therapeutics' Anaphylm Gets Rejected | 1.190 | AFX News | PETAH TIKVA (dpa-AFX) - This week's biotech landscape witnessed key FDA meetings being scheduled, IND clearances, complete response letters, sale of oncology assets and clinical trial data readouts... ► Artikel lesen | |
| 03.02. | Sangamo gene therapy shows positive kidney function results in Fabry disease | 56 | Investing.com |
1 2 Weiter >>
32 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 79,25 | -3,29 % | Vorsicht bei BioNTech! Novo Nordisk gibt Gas! Kaufchance bei Vidac Pharma Aktie! | Kaufstimmung bei Novo Nordisk. Die Aktie des dänischen Pharmagiganten scheint den seit Sommer 2024 begonnenen Abwärtstrend zu verlassen. Anleger reagieren auf positive Meldungen. Bei Vidac Pharma ist... ► Artikel lesen | |
| EVOTEC | 5,305 | +0,47 % | EQS-NVR: Evotec SE: Veröffentlichung der Gesamtzahl der Stimmrechte nach § 41 WpHG mit dem Ziel der europaweiten Verbreitung | EQS Gesamtstimmrechtsmitteilung: Evotec SE
/ Veröffentlichung der Gesamtzahl der Stimmrechte
Evotec SE: Veröffentlichung der Gesamtzahl der Stimmrechte nach § 41 WpHG mit dem... ► Artikel lesen | |
| BB BIOTECH | 47,100 | -1,57 % | BB Biotech: "Wachstumsstarke Mid- und Small Caps bilden das Rückgrat unseres Portfolios": Mehr Einzelwerte und ... | ||
| MEDIGENE | 0,027 | -6,21 % | PTA-Adhoc: Medigene AG: Widerruf der Zulassung der Aktien der Medigene AG zum Handel im regulierten Markt | DJ PTA-Adhoc: Medigene AG: Widerruf der Zulassung der Aktien der Medigene AG zum Handel im regulierten Markt
Veröffentlichung von Insiderinformationen gemäß Artikel 17 MAR Schlagwort(e): Sonstiges... ► Artikel lesen | |
| QIAGEN | 30,815 | -2,62 % | DEUTSCHE BANK RESEARCH stuft QIAGEN NV auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat die Einstufung für Qiagen mit einem Kursziel von 43 US-Dollar auf "Buy" belassen. Das Interesse der Investoren habe vor allem dem Tuberkulosetest... ► Artikel lesen | |
| MODERNA | 39,675 | +0,18 % | Moderna, Inc.: Moderna and Merck Present 5-Year Data for Intismeran Autogene in Combination With KEYTRUDA in Patients With High-Risk Stage III/IV Melanoma Following Complete Resection at the 2026 ASCO Annual Meeting | At a median 5-year (60.3 months) planned follow-up of the Phase 2b KEYNOTE-942/mRNA-4157-P201 study, intismeran autogene in combination with KEYTRUDA demonstrated a 49% reduction in the risk of recurrence... ► Artikel lesen | |
| PAION | 0,065 | -21,21 % | XFRA MISTRADE ANTRAG IN ISIN DE000A3E5EG5 WURDE STATTGEGEBEN | Dem Mistrade-Antrag wurde stattgegeben. Folgende Geschaefte in der ISIN DE000A3E5EG5, Wertpapier-Name: PAION AG INH O.N., wurden aufgehoben:ISIN Datum Zeit Volumen PreisDE000A3E5EG5 02.02.2026 08:13:40 300 0... ► Artikel lesen | |
| VALNEVA | 2,500 | -0,56 % | Original-Research: Valneva SE (von First Berlin Equity Research GmbH): Buy | Original-Research: Valneva SE - von First Berlin Equity Research GmbH
28.05.2026 / 17:08 CET/CEST
Veröffentlichung einer Research, übermittelt durch EQS News - ein Service der EQS Group.
Für den... ► Artikel lesen | |
| AMGEN | 283,80 | +0,30 % | Mesoblast: Während Amgen und Novo Nordisk den Biotech-Hype treiben, arbeitet dieser Zelltherapie-Player am operativen Durchbruch | ||
| EPIGENOMICS | 0,870 | 0,00 % | PTA-HV: Epigenomics AG: Einladung zur ordentlichen Hauptversammlung | DJ PTA-HV: Epigenomics AG: Einladung zur ordentlichen Hauptversammlung
Hauptversammlung gemäß -- 121 Abs. 4a AktG
Epigenomics AG: Einladung zur ordentlichen Hauptversammlung
Heidelberg... ► Artikel lesen | |
| NOVAVAX | 9,230 | +2,10 % | Novavax, Inc. (NVAX) Is a Trending Stock: Facts to Know Before Betting on It | ||
| STRYKER | 258,90 | +0,54 % | Stryker bringt das Pangea-Plattensystem auf den Markt und schließt den ersten Eingriff in Europa ab | Stryker (NYSE:SYK), ein weltweit führendes Unternehmen im Bereich Medizintechnik, gab heute die Markteinführung seines Pangea-Plattensystems in Europa bekannt, einer Plattensystem-Plattform zur Behandlung... ► Artikel lesen | |
| BIOGEN | 164,90 | -0,23 % | UCB and Biogen Announce Publication in The Lancet of Positive Dapirolizumab Pegol (DZP) Phase 3 Study Results in Systemic Lupus Erythematosus | Publication adds to previously reported findings from Phase 3 PHOENYCS GO study, demonstrating statistically significant improvement in disease activity at Week 48 with dapirolizumab pegol plus standard... ► Artikel lesen | |
| BIOFRONTERA | 2,530 | +3,69 % | PTA-News: Biofrontera AG: Biofrontera berichtet über die Ergebnisse des ersten Quartals 2026 | DJ PTA-News: Biofrontera AG: BIOFRONTERA BERICHTET ÜBER DIE ERGEBNISSE DES ERSTEN QUARTALS 2026
Unternehmensmitteilung für den Kapitalmarkt
Biofrontera AG: BIOFRONTERA BERICHTET ÜBER... ► Artikel lesen | |
| HEIDELBERG PHARMA | 2,780 | +1,46 % | EQS-HV: Heidelberg Pharma AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in Ladenburg mit dem Ziel der europaweiten Verbreitung gemäß §121 AktG | EQS-News: Heidelberg Pharma AG
/ Bekanntmachung der Einberufung zur Hauptversammlung
Heidelberg Pharma AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in... ► Artikel lesen |
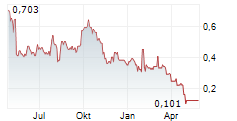
Das Unternehmen SANGAMO THERAPEUTICS INC kann der Branche Biotechnologie zugeordnet werden. Die relative Kursveränderung beträgt aktuell -100,00 % bei einem Aktienkurs von 0,126 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu SANGAMO THERAPEUTICS INC finden Sie auf Wallstreet Online.