- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| Di | Pasithea receives orphan drug status for ALS treatment candidate | 1 | Investing.com | ||
| Di | Pasithea Therapeutics Announces Orphan Drug Designation by FDA of PAS-004 for Treatment of Amyotrophic Lateral Sclerosis (ALS) | 132 | GlobeNewswire (Europe) | MIAMI, June 02, 2026 (GLOBE NEWSWIRE) -- Pasithea Therapeutics Corp. (NASDAQ: KTTA) ("Pasithea" or the "Company"), a clinical-stage biotechnology company developing PAS-004, a next-generation macrocyclic... ► Artikel lesen | |
| 04.05. | Pasithea Therapeutics Corp. - 8-K, Current Report | - | SEC Filings | ||
| PASITHEA THERAPEUTICS Aktie jetzt für 0€ handeln | |||||
| 04.05. | Pasithea Therapeutics appoints chief medical officer | 1 | Seeking Alpha | ||
| 04.05. | Pasithea names Kartik Krishnan as chief medical officer | 1 | Investing.com | ||
| 04.05. | Pasithea Therapeutics Announces Appointment of Kartik Krishnan, M.D., Ph.D. as Chief Medical Officer | 135 | GlobeNewswire (Europe) | MIAMI, May 04, 2026 (GLOBE NEWSWIRE) -- Pasithea Therapeutics Corp. (NASDAQ: KTTA) ("Pasithea" or the "Company"), a clinical-stage biotechnology company developing PAS-004, a next-generation macrocyclic... ► Artikel lesen | |
| 20.04. | FDA grants rare pediatric disease status to Pasithea's PAS-004 | 6 | Investing.com | ||
| 20.04. | Pasithea Therapeutics Announces Grant of Rare Pediatric Disease Designation (RPDD) by FDA to PAS-004 for Treatment of Neurofibromatosis Type 1 (NF1) | 234 | GlobeNewswire (Europe) | MIAMI, April 20, 2026 (GLOBE NEWSWIRE) -- Pasithea Therapeutics Corp. (NASDAQ: KTTA) ("Pasithea" or the "Company"), a clinical-stage biotechnology company developing PAS-004, a next-generation macrocyclic... ► Artikel lesen | |
| 01.04. | Pasithea Therapeutics Announces Grant of Fast Track Designation by FDA to PAS-004 for Treatment of Neurofibromatosis Type 1 (NF1) Associated Plexiform Neurofibromas (PN) Causing Significant Morbidity | 138 | GlobeNewswire (Europe) | MIAMI, April 01, 2026 (GLOBE NEWSWIRE) -- Pasithea Therapeutics Corp. (NASDAQ: KTTA) ("Pasithea" or the "Company"), a clinical-stage biotechnology company developing PAS-004, a next-generation macrocyclic... ► Artikel lesen | |
| 20.02. | Pasithea Therapeutics Corp. - 8-K, Current Report | 4 | SEC Filings | ||
| 30.01. | Pasithea Therapeutics Corp. - S-8, Securities to be offered to employees in employee benefit plans | 1 | SEC Filings | ||
| 13.01. | Pasithea Therapeutics Provides Outlook on PAS-004 Clinical Programs and Data Release Timelines | 205 | GlobeNewswire (Europe) | MIAMI, Jan. 13, 2026 (GLOBE NEWSWIRE) -- Pasithea Therapeutics Corp. (NASDAQ: KTTA) ("Pasithea" or the "Company"), a clinical-stage biotechnology company developing PAS-004, a next-generation oral... ► Artikel lesen | |
| 15.12.25 | Pasithea Therapeutics Corp. - 8-K, Current Report | - | SEC Filings | ||
| 21.11.25 | Pasithea Therapeutics Announces Positive PAS-004 Tablet Pharmacokinetic (PK) Data in Ongoing Phase 1/1b Trial in Adult NF1 Patients | 347 | GlobeNewswire (Europe) | -- Tablet PK exposure increases proportionally with an increase in dose -- -- More favorable PK properties in tablets enable a lower dose to achieve the same exposure as the capsule formulation, with... ► Artikel lesen | |
| 20.11.25 | Pasithea Therapeutics Announces Positive Phase 1 Data Including Partial Response, Demonstrating Monotherapy Clinical Activity and Favorable Safety Profile for PAS-004 in Advanced Cancer Study | 248 | GlobeNewswire (Europe) | -- Evidence of Monotherapy Activity: Partial response observed in a MEK-rechallenge 3rd-line melanoma patient with BRAF V600E mutation who remains on trial for more than 11 months -- -- A second MEK-rechallenge... ► Artikel lesen | |
| 04.11.25 | Pasithea Therapeutics Announces Activation of Clinical Trial Site at University of Alabama at Birmingham for Ongoing Phase 1/1b Trial of PAS-004 in Adult NF1 Patients | 178 | GlobeNewswire (Europe) | MIAMI, Nov. 04, 2025 (GLOBE NEWSWIRE) -- Pasithea Therapeutics Corp. (Nasdaq: KTTA) ("Pasithea" or the "Company"), a clinical-stage biotechnology company developing PAS-004, a next-generation macrocyclic... ► Artikel lesen | |
| 16.09.25 | Pasithea Therapeutics Announces Activation of Clinical Trial Sites in South Korea for Phase 1/1b Trial of PAS-004 in Adult NF1 Patients | 198 | GlobeNewswire (Europe) | MIAMI, Sept. 16, 2025 (GLOBE NEWSWIRE) -- Pasithea Therapeutics Corp. (NASDAQ: KTTA) ("Pasithea" or the "Company"), a clinical-stage biotechnology company developing PAS-004, a next-generation macrocyclic... ► Artikel lesen | |
| 08.09.25 | Pasithea Therapeutics Announces Enrollment of Cohort 2 following Positive Safety Review Committee (SRC) Recommendation for its Ongoing Phase 1/1b Clinical Trial of PAS-004 in Adult NF1 Patients | 188 | GlobeNewswire (Europe) | -- Recommendation that trial escalate to next dose level of 8mg tablet -- -- Initial interim clinical data from first two cohorts expected in Q1 2026 -- MIAMI, Sept. 08, 2025 (GLOBE NEWSWIRE)... ► Artikel lesen | |
| 31.07.25 | Pasithea Therapeutics Completes Enrollment and Initial Dosing of First Cohort from its Phase 1/1b Clinical Trial of PAS-004 in Adult NF1 Patients | 470 | GlobeNewswire (Europe) | MIAMI, July 31, 2025 (GLOBE NEWSWIRE) -- Pasithea Therapeutics Corp. (NASDAQ: KTTA) ("Pasithea" or the "Company"), a clinical-stage biotechnology company developing PAS-004, a next-generation macrocyclic... ► Artikel lesen |
19 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| OCUGEN | 1,118 | +0,18 % | Ocugen, Inc. - 8-K, Current Report | ||
| ABIVAX | 87,00 | -0,63 % | Abivax mit Daten: Erst Rakete, dann Absturz - erhöht Obefazimod das Krebsrisiko? | Die Aktie von Abivax hat am Montagabend genau die Nachricht geliefert, auf die Investoren seit Monaten gewartet hatten. Das französische Biotech-Unternehmen präsentierte herausragende Topline-Ergebnisse... ► Artikel lesen | |
| ADAPTIVE BIOTECHNOLOGIES | 14,600 | -0,41 % | Adaptive Biotechnologies Reports Fourth Quarter and Full Year 2025 Financial Results | SEATTLE, Feb. 05, 2026 (GLOBE NEWSWIRE) -- Adaptive Biotechnologies Corporation ("Adaptive Biotechnologies") (Nasdaq: ADPT), a commercial stage biotechnology company that aims to translate the genetics... ► Artikel lesen | |
| GENPREX | 0,655 | +1,22 % | Genprex, Inc.: Genprex Strengthens Intellectual Property Portfolio with Japanese and EU Patents for Reqorsa Gene Therapy in Combination with Immunotherapies to Treat Cancer | Japanese Patent Covers Acclaim-3 Clinical Trial Combining REQORSA Gene Therapy with Genentech, Inc.'sTecentriq®
AUSTIN, Texas, Feb. 23, 2026 /PRNewswire/ -- Genprex... ► Artikel lesen | |
| REZOLUTE | 3,800 | +2,70 % | Rezolute, Inc.: Rezolute Announces Four Presentations at the Upcoming Annual Meeting of the Endocrine Society | ||
| SPRUCE BIOSCIENCES | 49,000 | -2,84 % | Spruce Biosciences, Inc.: Spruce Biosciences Announces Data Presentation at the 18th International MPS Symposium | Nicole Muschol, M.D. to Present Long-Term Data on Tralesinidase Alfa (TA-ERT) in Sanfilippo Syndrome Type B (MPS IIIB) Spruce Biosciences, Inc. (Nasdaq: SPRB), a late-stage biopharmaceutical... ► Artikel lesen | |
| UNICYCIVE THERAPEUTICS | 6,920 | 0,00 % | Unicycive Therapeutics, Inc. - 8-K, Current Report | ||
| DAMORA THERAPEUTICS | 21,040 | +0,14 % | Damora Therapeutics, Inc.: Damora Therapeutics Appoints Biotechnology Leader Jennifer Jarrett as President and Chief Executive Officer; Adds Two Industry Veterans to Board of Directors | -- Ms. Jarrett brings nearly three decades of executive leadership in biotech, tech and finance -- -- Biotech leaders Dr. Cameron Turtle and Mike Landsittel appointed to Board of Directors -- -- Peter... ► Artikel lesen | |
| FIBROBIOLOGICS | 1,015 | -1,46 % | FibroBiologics, Inc.: FibroBiologics Announces First Patients Dosed in Phase 1/2 Clinical Trial Evaluating CYWC628 for the Treatment of Diabetic Foot Ulcers | HOUSTON, June 03, 2026 (GLOBE NEWSWIRE) -- FibroBiologics, Inc. (Nasdaq: FBLG) ("FibroBiologics"), a clinical-stage biotechnology company with 270+ patents issued and pending with a focus on the development... ► Artikel lesen | |
| PRAXIS PRECISION MEDICINES | 254,00 | 0,00 % | Praxis Precision Medicines, Inc.: Praxis Precision Medicines Provides Vormatrigine Program Update | POWER1 Study in highly refractory patients with focal onset seizures did not meet its primary success measure Secondary measure, the 50% response rate, was met and seizure reduction during the second... ► Artikel lesen | |
| EDGEWISE THERAPEUTICS | 35,590 | 0,00 % | Servier to Acquire Edgewise Therapeutics' Muscular Dystrophy Business | Acquisition supports Servier's strategic ambition in rare neurology for patients with high unmet medical needs.Transaction includes sevasemten, a late-stage investigational treatment for... ► Artikel lesen | |
| JADE BIOSCIENCES | 16,940 | 0,00 % | Jade Biosciences Announces Positive Interim Phase 1 Results for JADE101 Demonstrating Potential Best-in-Class Profile and Every-12-Week Dosing in IgA Nephropathy | ~70% IgA reductions sustained at 12 weeks after a single dose of JADE101>70% IgA reductions projected at steady state with a single subcutaneous maintenance injection every 12 weeksIgA-lowering effect... ► Artikel lesen | |
| QIAGEN | 31,920 | -2,18 % | DEUTSCHE BANK RESEARCH stuft QIAGEN NV auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat Qiagen mit einem Kursziel von 43 US-Dollar auf "Buy" belassen. Das Feedback aus Gesprächen mit etlichen US-Laborexperten deute darauf hin, dass... ► Artikel lesen | |
| APOGEE THERAPEUTICS | 86,86 | -0,07 % | Weekly Buzz: AstraZeneca, JNJ Get FDA Nod; Eli Lilly To Acquire Curevo; APGE Advances Zumilokibart | NEW BRUNSWICK (dpa-AFX) - This week in the biotech space, there were regulatory approvals across the U.S. and Europe, collaborations, acquisitions, and positive data readouts spanning multiple... ► Artikel lesen | |
| ADMA BIOLOGICS | 7,970 | 0,00 % | ADMA BIOLOGICS, INC. - 8-K, Current Report |
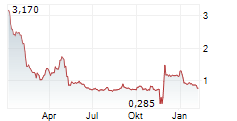
PASITHEA THERAPEUTICS CORP gehört der Branche Biotechnologie an. Mit einer aktuellen Kursveränderung von +0,004 (+0,63 %) liegt der Kurs bei 0,589 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu PASITHEA THERAPEUTICS CORP finden Sie auf Wallstreet Online.