| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
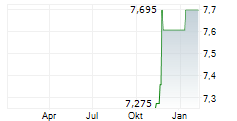
| 7,700 | 7,900 | 13:02 | |
| 7,600 | 7,900 | 22.05. |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| SHANGHAI HENLIUS BIOTECH Aktie jetzt für 0€ handeln | |||||
| Fr | HENLIUS (02696): VOLUNTARY ANNOUNCEMENT - SERPLULIMAB INJECTION (TRADE NAME IN CHINESE MAINLAND: HANSIZHUANG; TRADE NAME IN THE EUROPEAN UNION: HETRONIFLY) ... | - | HKEx | ||
| Fr | HENLIUS (02696): VOLUNTARY ANNOUNCEMENT - THE FIRST PATIENT HAS BEEN DOSED IN A PHASE 1 CLINICAL STUDY OF HLX316 FOR INJECTION (B7-H3-TARGETING SIALIDASE ... | 1 | HKEx | ||
| Fr | Alligator Bioscience Comments on Henlius Update on Long-Term Follow-Up Data for HLX22 | 231 | ACCESS Newswire | LUND, SE / ACCESS Newswire / May 22, 2026 / Alligator Bioscience (Nasdaq Stockholm:ATORX) today comments on a recent update shared by Shanghai Henlius Biotech, Inc. regarding long-term follow-up observations... ► Artikel lesen | |
| Do | HENLIUS (02696): VOLUNTARY ANNOUNCEMENT - THE FIRST PATIENT IN AN EU COUNTRY (SPAIN) HAS BEEN DOSED IN AN INTERNATIONAL MULTICENTRE PHASE 2 CLINICAL STUDY ... | 2 | HKEx | ||
| Do | HENLIUS (02696): VOLUNTARY ANNOUNCEMENT - THE PHASE 1 CLINICAL TRIAL OF HLX3902 INJECTION (STEAP1XCD3XCD28 TRISPECIFIC ANTIBODY) FOR THE TREATMENT OF ... | - | HKEx | ||
| Do | HENLIUS (02696): VOLUNTARY ANNOUNCEMENT - THE INVESTIGATIONAL NEW DRUG APPLICATION FOR THE PHASE 1 CLINICAL TRIAL OF HLX48 FOR INJECTION (AN ANTIBODY-DRUG ... | - | HKEx | ||
| Di | HENLIUS (02696): POLL RESULTS OF THE RESOLUTIONS PROPOSED AT THE ANNUAL GENERAL MEETING HELD ON TUESDAY, 19 MAY 2026 | 1 | HKEx | ||
| 13.05. | HENLIUS (02696): VOLUNTARY ANNOUNCEMENT - THE PHASE 1 CLINICAL TRIAL OF HLX48 FOR INJECTION (AN ANTIBODY-DRUG CONJUGATE TARGETING EGFR AND C-MET) FOR ... | 2 | HKEx | ||
| 13.05. | HENLIUS (02696): VOLUNTARY ANNOUNCEMENT - THE FIRST PATIENT HAS BEEN DOSED IN A PHASE 1 CLINICAL STUDY OF HLX97 (KAT6A/B SMALL MOLECULE INHIBITOR) IN ... | - | HKEx | ||
| 11.05. | HENLIUS HLX05-N Receives FDA Approval for Phase 1 Clinical Trial IND for Metastatic Colorectal Cancer Treatment | 1 | AASTOCKS | ||
| 04.05. | HENLIUS HLX43 for Injection Approved in Japan to Conduct Relevant Clinical Trials | 1 | AASTOCKS | ||
| 04.05. | HENLIUS (02696): VOLUNTARY ANNOUNCEMENT - AN INTERNATIONAL MULTICENTER PHASE 2/3 CLINICAL TRIAL OF HLX43 FOR INJECTION (AN ANTI-PD-L1 ANTIBODY-DRUG CONJUGATE) ... | 1 | HKEx | ||
| 30.04. | Organon & Co.: Europäische Kommission erteilt Marktzulassung für POHERDY (Pertuzumab) von Henlius und Organon, das erste in Europa zugelassene Biosimilar zu PERJETA (Pertuzumab) | 471 | Business Wire | Shanghai Henlius Biotech, Inc. (2696.HK) und Organon (NYSE: OGN) gaben heute bekannt, dass die Europäische Kommission die Zulassung für POHERDY (Pertuzumab) 420 mg/14 ml Injektion zur intravenösen... ► Artikel lesen | |
| 30.04. | HENLIUS HLX3901 Injection Phase 1 Clinical Study Completes First Patient Dosing in China | 1 | AASTOCKS | ||
| 30.04. | HENLIUS (02696): VOLUNTARY ANNOUNCEMENT - THE FIRST PATIENT HAS BEEN DOSED IN A PHASE 1 CLINICAL STUDY OF HLX3901 INJECTION (DLL3XDLL3XCD3XCD28 TETRA-SPECIFIC ... | - | HKEx | ||
| 29.04. | Henlius And Organon Get EU Green Light For First Pertuzumab Biosimilar | 1 | RTTNews | ||
| 29.04. | Organon & Co.: European Commission (EC) Approves Henlius and Organon's POHERDY (pertuzumab), the First Approved Biosimilar to PERJETA (pertuzumab) in Europe | 363 | Business Wire | Shanghai Henlius Biotech, Inc. (2696.HK), and Organon (NYSE: OGN) today announced the European Commission (EC) has granted marketing authorization for POHERDY (pertuzumab) 420 mg/14 mL injection for... ► Artikel lesen | |
| 28.04. | HENLIUS (02696): INSIDE INFORMATION ANNOUNCEMENT - EUROPEAN COMMISSION (EC) APPROVED HLX11 (PERTUZUMAB, TRADE NAME IN THE UNITED STATES AND EUROPE: POHERDY) ... | - | HKEx | ||
| 24.04. | HENLIUS (02696): VOLUNTARY ANNOUNCEMENT - THE PHASE 2/3 CLINICAL TRIAL OF PIMURUTAMAB HLX07 (RECOMBINANT ANTI-EGFR HUMANISED MONOCLONAL ANTIBODY INJECTION) ... | 1 | HKEx | ||
| 24.04. | HENLIUS (02696): PROXY FORM FOR THE ANNUAL GENERAL MEETING TO BE HELD ON TUESDAY, 19 MAY 2026 (OR ANY ADJOURNMENT THEREOF) | - | HKEx |
1 2 3 4 5 Weiter >>
89 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| SUMMIT THERAPEUTICS | 16,620 | 0,00 % | This Summit Therapeutics Analyst Is No Longer Bullish; Here Are Top 2 Downgrades For Friday | ||
| CG ONCOLOGY | 64,25 | 0,00 % | Wolfe Research startet Coverage für CG Oncology mit "Peerperform" | ||
| QIAGEN | 30,290 | +0,65 % | QIAGEN N.V.: QIAGEN to advance AI-driven drug discovery with graph-based AI and curated bioinformatics knowledge with NVIDIA | Collaboration integrates NVIDIA accelerated computing and the NVIDIA BioNeMo platform with QIAGEN Digital Insights' curated biomedical knowledge bases Goal to advance how disease mechanisms... ► Artikel lesen | |
| TANGO THERAPEUTICS | 20,240 | 0,00 % | Tango Therapeutics, Inc.: Tango Therapeutics Reports First Quarter 2026 Financial Results and Provides Business Highlights | First clinical data from PRMT5/RAS(ON) combination trial to be presented in 2026 Cash position of $380 million as of March 31, 2026, with runway into 2028 beyond anticipated key data inflection points... ► Artikel lesen | |
| PRAXIS PRECISION MEDICINES | 343,26 | -0,09 % | Praxis Precision Medicines, Inc.: Praxis Precision Medicines Provides Corporate Update and Reports First Quarter 2026 Financial Results | FDA accepted the new drug application (NDA) for ulixacaltamide in Essential Tremor with a PDUFA target action date of January 29, 2027, and the NDA for relutrigine, with priority review, in SCN2A... ► Artikel lesen | |
| COGENT BIOSCIENCES | 32,640 | -0,09 % | Cogent Biosciences: Aktie legt vor wichtigen Studiendaten um 4 % zu | ||
| GEOVAX LABS | 3,630 | -0,27 % | EQS-News: GeoVax, Inc.: GeoVax Comments on Escalating Bundibugyo Ebola Outbreak and Growing Need for Flexible Biodefense Vaccine Platforms | EQS-News: GeoVax, Inc.
/ Key word(s): Science
GeoVax Comments on Escalating Bundibugyo Ebola Outbreak and Growing Need for Flexible Biodefense Vaccine Platforms 20.05.2026 / 16:06... ► Artikel lesen | |
| METAVIA | 3,850 | 0,00 % | MetaVia: Präklinische Studie zu Vanoglipel untermauert Potenzial bei Lebererkrankungen | ||
| IMMUNEERING | 5,390 | 0,00 % | Immuneering Corporation: Immuneering Reports 17.3 Months Median Overall Survival in First-Line Metastatic Pancreatic Cancer Patients Treated with Atebimetinib Plus Chemotherapy | - Detailed data from 55 patients to be shared in an oral presentation at the ASCO Annual Meeting on June 1, 2026 - - Tolerability profile consistent with prior updates: only two categories of Grade... ► Artikel lesen | |
| RELAY THERAPEUTICS | 13,535 | +0,04 % | Relay Therapeutics, Inc.: Relay Therapeutics Announces Pricing of Public Offering of Common Stock | CAMBRIDGE, Mass., May 20, 2026 (GLOBE NEWSWIRE) -- Relay Therapeutics, Inc. (Nasdaq: RLAY), a clinical-stage, small molecule precision medicine company developing potentially life-changing therapies... ► Artikel lesen | |
| TAYSHA GENE THERAPIES | 5,490 | 0,00 % | Taysha Gene Therapies, Inc.: Taysha Gene Therapies Reports First Quarter 2026 Financial Results and Provides Corporate Update | Reaffirmed FDA alignment on TSHA-102 BLA pathway, including pivotal trial design, endpoints and potential to submit for approval based on REVEAL pivotal trial 6-month interim analysis, following... ► Artikel lesen | |
| IMMUNOVANT | 34,140 | -0,03 % | Immunovant Inc.: Immunovant Provides Corporate Updates and Reports Financial Results for the Fourth Quarter and Fiscal Year Ended March 31, 2026 | IMVT-1402 showed clinically meaningful response rates of 72.7% ACR20, 54.5% ACR50 and 35.8% ACR70 at Week 16 in the open label period of its trial in difficult-to-treat rheumatoid arthritis (D2T RA)... ► Artikel lesen | |
| BEAM THERAPEUTICS | 28,640 | 0,00 % | Beam Therapeutics presents AATD gene therapy trial data at ATS | ||
| INHIBRX BIOSCIENCES | 101,45 | +0,13 % | Inhibrx Biosciences, Inc.: Inhibrx Reports First Quarter 2026 Financial Results | SAN DIEGO, May 14, 2026 /PRNewswire/ -- Inhibrx Biosciences, Inc. (Nasdaq: INBX) ("Inhibrx" or the "Company") today reported financial results for the first quarter... ► Artikel lesen | |
| RECURSION PHARMACEUTICALS | 3,005 | -0,17 % | Morgan Stanley Raises its Price Target on Recursion (RXRX) |
Das Unternehmen SHANGHAI HENLIUS BIOTECH INC kann der Branche Biotechnologie zugeordnet werden. Der aktuelle Kurs liegt bei 7,850 Euro und damit +1,29 % im Plus.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu SHANGHAI HENLIUS BIOTECH INC finden Sie auf Wallstreet Online.