- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 28.05. | Silexion Therapeutics Corp - S-8, Securities to be offered to employees in employee benefit plans | 13 | SEC Filings | ||
| 26.05. | Silexion Therapeutics announces 1-for-10 reverse share split | 1 | Seeking Alpha | ||
| 26.05. | Silexion announces 1-for-10 reverse stock split effective Thursday | 1 | Investing.com | ||
| 26.05. | Silexion Therapeutics Corp - 8-K, Current Report | 2 | SEC Filings | ||
| SILEXION THERAPEUTICS Aktie jetzt für 0€ handeln | |||||
| 26.05. | Silexion: Aktienzusammenlegung im Verhältnis 1 zu 10 wird am Donnerstag wirksam | 1 | Investing.com Deutsch | ||
| 26.05. | Silexion Therapeutics Corp: Silexion Therapeutics Announces 1-for-10 Reverse Share Split | 259 | GlobeNewswire (Europe) | Grand Cayman, May 26, 2026 (GLOBE NEWSWIRE) -- Silexion Therapeutics Corp. (NASDAQ: SLXN) ("Silexion" or the "Company"), a clinical-stage biotechnology company pioneering RNA interference (RNAi) therapies... ► Artikel lesen | |
| 19.05. | Silexion Therapeutics Corp - 10-K/A, Annual Report | 2 | SEC Filings | ||
| 18.05. | Silexion Therapeutics GAAP EPS of -$0.85 beats by $0.44 | 1 | Seeking Alpha | ||
| 18.05. | Silexion Therapeutics Corp: Silexion Therapeutics Reports First Quarter 2026 Financial Results and Provides Business Update | 327 | GlobeNewswire (Europe) | Israeli Ministry of Health approved the initiation of Silexion's Phase 2/3 clinical trial of SIL204 in locally advanced pancreatic cancer Clinical Trial Application subsequently submitted to Germany... ► Artikel lesen | |
| 15.05. | Silexion Therapeutics Corp - 10-Q, Quarterly Report | 1 | SEC Filings | ||
| 15.05. | Silexion beschafft 1 Million Dollar über Optionsscheine | 2 | Investing.com Deutsch | ||
| 15.05. | Silexion Therapeutics Corp: Silexion Therapeutics Announces Exercise of Warrants for $1 Million Gross Proceeds | 2 | GlobeNewswire (USA) | ||
| 14.05. | Silexion reports preclinical data on SIL204 in pancreatic cancer | 3 | Investing.com | ||
| 14.05. | Silexion Therapeutics Corp: Silexion Therapeutics Reports Positive Preliminary Immunotherapy Findings for SIL204 in KRAS-Driven Pancreatic Cancer | 164 | GlobeNewswire (Europe) | Grand Cayman, Cayman Islands, May 14, 2026 (GLOBE NEWSWIRE) -- Silexion Therapeutics Corp. (NASDAQ: SLXN) ("Silexion" or the "Company"), a clinical-stage biotechnology company pioneering RNA interference... ► Artikel lesen | |
| 13.05. | Silexion Therapeutics Corp - 8-K, Current Report | 2 | SEC Filings | ||
| 13.05. | Silexion begins clinical manufacturing of SIL204 for phase 2/3 trial | 4 | Investing.com | ||
| 13.05. | Silexion Therapeutics Corp: Silexion Therapeutics Announces Initiation of GMP Clinical Supply Manufacturing of SIL204 with Leading Global CDMO, and New Approval of Phase 2/3 Trial From Tel Aviv Sourasky Medical Center | 211 | GlobeNewswire (Europe) | Silexion advances toward Phase 2/3 trial in KRAS-driven pancreatic cancer, as SIL204 begins GMP clinical manufacturing in collaboration with global leading manufacturing partner Catalent at its European... ► Artikel lesen | |
| 05.05. | Silexion Therapeutics Corp - 8-K, Current Report | - | SEC Filings | ||
| 28.04. | Silexion Therapeutics Corp: Silexion Therapeutics Announces Successful Submission of Phase 2/3 Clinical Trial Application to Germany's BfArM for SIL204 in KRAS-Driven Locally Advanced Pancreatic Cancer | 457 | GlobeNewswire (Europe) | Grand Cayman, Cayman Islands, April 28, 2026 (GLOBE NEWSWIRE) -- Silexion Therapeutics Corp. (NASDAQ: SLXN) ("Silexion" or the "Company"), a clinical-stage biotechnology company pioneering RNA interference... ► Artikel lesen | |
| 28.04. | Silexion Therapeutics Corp - 8-K, Current Report | 1 | SEC Filings |
1 2 3 Weiter >>
43 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 77,05 | -1,34 % | BioNTech und BMS melden neue Daten für Lungenkrebskandidat Pumitamig | BioNTech SE und Bristol Myers Squibb haben auf dem ASCO-Jahreskongress in Chicago erste globale Phase-2-Daten für den bispezifischen Immunmodulator Pumitamig in Kombination mit Chemotherapie bei fortgeschrittenem... ► Artikel lesen | |
| AMGEN | 306,30 | +0,97 % | Während Merck, Amgen, Novo Nordisk und Fresenius Milliarden bewegen, wächst die Fantasie im Zelltherapie-Sektor | ||
| NOVAVAX | 8,080 | -1,70 % | NOVAVAX INC: Stabilität vor der nächsten Bewegung | ||
| BIOGEN | 171,56 | +1,19 % | Biogen's Salanersen Wins FDA Breakthrough Therapy Designation For Spinal Muscular Atrophy Treatment | WESTON (dpa-AFX) - Thursday, Biogen Inc. (BIIB) announced that the U.S. Food and Drug Administration has granted salanersen Breakthrough Therapy Designation for the treatment of spinal muscular... ► Artikel lesen | |
| VIKING THERAPEUTICS | 24,650 | -0,20 % | XETR NEW INSTRUMENTS AVAILABLE ON XETRA - 04.06.2026 | Folgende Wertpapiere werden in den Handel an folgenden Handelsplätzen (MIC Code) im Quotation Board an der Frankfurter Wertpapierbörse (FWB) aufgenommen. Die Notierung an anderen Handelsplätzen im Quotation... ► Artikel lesen | |
| INTELLIA THERAPEUTICS | 11,240 | -4,26 % | Intellia Therapeutics, Inc. (NTLA) Buy Rating Reiterated Despite Share Offering Dilution | ||
| TEMPUS AI | 40,195 | -0,22 % | Tempus AI gründet Open-Source-Konsortium für digitale Pathologie | ||
| BIOCRYST PHARMACEUTICALS | 7,232 | -0,85 % | BioCryst Ireland, a Neopharmed Gentili company: Hereditary angioedema, a European initiative to shine a spotlight on a still under-recognized condition | Ahead of World HAE Day, BioCryst Ireland, a Neopharmed Gentili company, organised an international event in Milan bringing together leading experts from across Europe to raise awareness of... ► Artikel lesen | |
| BIOMARIN PHARMACEUTICAL | 49,500 | +0,49 % | BIOMARIN PHARMACEUTICAL INC - 8-K, Current Report | ||
| EXELIXIS | 45,530 | -0,40 % | Exelixis präsentiert positive Studiendaten zu Cabometyx bei neuroendokrinen Tumoren | ||
| SAREPTA THERAPEUTICS | 13,800 | +0,88 % | Improving Profitability Supports Sarepta Therapeutics Inc. (SRPT) as One of the Most Shorted Small-Cap Stocks to Buy | ||
| PACIFIC BIOSCIENCES OF CALIFORNIA | 1,235 | -1,56 % | PACIFIC BIOSCIENCES OF CALIFORNIA, INC. - 8-K, Current Report | ||
| CARDIOL THERAPEUTICS | 0,989 | +2,17 % | Original-Research: Cardiol Therapeutics Inc (von First Berlin Equity Research GmbH): BUY | Original-Research: Cardiol Therapeutics Inc - from First Berlin Equity Research GmbH
30.04.2026 / 13:07 CET/CEST
Dissemination of a Research, transmitted by EQS News - a service of EQS Group.
The... ► Artikel lesen | |
| VAXART | 0,380 | 0,00 % | Vaxart, Inc.: Vaxart Files Definitive Proxy Statement and Mails Letter to Shareholders | Emphasizes Importance of Experienced Biotech Leadership as Vaxart Advances Clinical and Strategic Priorities with Multiple Value-Creating Milestones Ahead Highlights Significant Risk of Replacing... ► Artikel lesen | |
| ATAIBECKLEY | 3,400 | -0,58 % | Wall Street entdeckt Psychedelika - steht Emyria vor einer Neubewertung? |
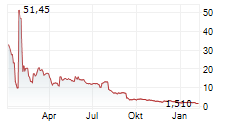
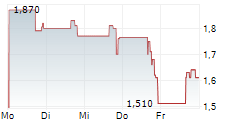
Das Unternehmen SILEXION THERAPEUTICS CORP kann der Branche Biotechnologie zugeordnet werden. Die relative Kursveränderung beträgt aktuell -2,36 % bei einem Aktienkurs von 4,560 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu SILEXION THERAPEUTICS CORP finden Sie auf Wallstreet Online.