| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
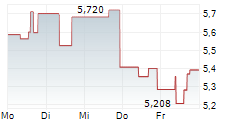
| 7,142 | 7,200 | 02.06. | |
| 7,120 | 7,178 | 02.06. |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 13.05. | BioCryst Pharmaceuticals, Inc.: BioCryst to Present New Real-World Evidence Underscoring the Ongoing Burden of Pediatric Hereditary Angioedema at ISPOR 2026 | 7 | GlobeNewswire (USA) | ||
| 13.05. | BioCryst Ireland, a Neopharmed Gentili company: Hereditary angioedema, a European initiative to shine a spotlight on a still under-recognized condition | 484 | PR Newswire | Ahead of World HAE Day, BioCryst Ireland, a Neopharmed Gentili company, organised an international event in Milan bringing together leading experts from across Europe to raise awareness of... ► Artikel lesen | |
| 12.05. | BioCryst Q1 2026 slides: dual HAE strategy targets $1.8B revenue | 7 | Investing.com | ||
| 07.05. | These Analysts Revise Their Forecasts On BioCryst Pharma After Q1 Earnings | 18 | Benzinga.com | ||
| 07.05. | Citizens raises BioCryst stock price target on Orladeyo growth outlook | 31 | Investing.com | ||
| 06.05. | BIOCRYST PHARMACEUTICALS INC - 10-Q, Quarterly Report | 2 | SEC Filings | ||
| BIOCRYST PHARMACEUTICALS Aktie jetzt für 0€ handeln | |||||
| 06.05. | BioCryst scraps eye candidate to focus on rare diseases | 13 | FierceBiotech | ||
| 06.05. | BioCryst Q1 2026 slides: acquisition charges mask 21% revenue growth | 11 | Investing.com | ||
| 06.05. | Full Transcript: BioCryst Pharma Q1 2026 Earnings Call | 4 | Benzinga.com | ||
| 06.05. | BioCryst Pharmaceuticals, Inc.: BioCryst Reports First Quarter 2026 Financial Results and Provides Business Update | 239 | GlobeNewswire (Europe) | - Q1 2026 ORLADEYO net revenue of $148.3 million (+11% y-o-y; +21% y-o-y on comparable basis excluding European revenue) - - Announced licensing agreement for European commercial rights to navenibart... ► Artikel lesen | |
| 05.05. | BioCryst Pharmaceuticals, Inc.: BioCryst Reports Inducement Grants Under Nasdaq Listing Rule 5635(c)(4) | 4 | GlobeNewswire (USA) | ||
| 04.05. | BioCryst vergibt Navenibart-Lizenz für Europa in 70-Millionen-Dollar-Deal | 10 | Investing.com Deutsch | ||
| 04.05. | BioCryst licenses navenibart rights in Europe for $70M upfront | 2 | Investing.com | ||
| 04.05. | BioCryst Pharmaceuticals, Inc.: BioCryst Announces European Licensing Agreement with Irish Affiliate of Neopharmed Gentili for Navenibart in Hereditary Angioedema | 296 | GlobeNewswire (Europe) | - BioCryst grants Irish affiliate of Neopharmed Gentili exclusive license to commercialize navenibart for hereditary angioedema in Europe - - BioCryst to receive $70M upfront, up to $275M in future... ► Artikel lesen | |
| 04.05. | BIOCRYST PHARMACEUTICALS INC - 8-K, Current Report | 1 | SEC Filings | ||
| 15.04. | BioCryst Pharmaceuticals, Inc.: BioCryst to Report First Quarter 2026 Financial Results on May 6 | 3 | GlobeNewswire (USA) | ||
| 06.04. | BioCryst ernennt Sandeep Menon zum neuen Forschungs- und Entwicklungschef | 17 | Investing.com Deutsch | ||
| 06.04. | BioCryst names Sandeep Menon as chief R&D officer | 4 | Investing.com | ||
| 06.04. | BioCryst Pharmaceuticals, Inc.: BioCryst Appoints Sandeep M. Menon Chief Research and Development Officer | 720 | GlobeNewswire (Europe) | - Experienced R&D leader with a track record of advancing innovative therapies from early development to global approval - RESEARCH TRIANGLE PARK, N.C., April 06, 2026 (GLOBE NEWSWIRE) -- BioCryst... ► Artikel lesen | |
| 03.04. | BioCryst Pharmaceuticals, Inc.: BioCryst Reports Inducement Grants Under Nasdaq Listing Rule 5635(c)(4) | 6 | GlobeNewswire (USA) |
1 2 3 4 5 Weiter >>
81 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 76,30 | -3,72 % | Vorsicht bei BioNTech! Novo Nordisk gibt Gas! Kaufchance bei Vidac Pharma Aktie! | Kaufstimmung bei Novo Nordisk. Die Aktie des dänischen Pharmagiganten scheint den seit Sommer 2024 begonnenen Abwärtstrend zu verlassen. Anleger reagieren auf positive Meldungen. Bei Vidac Pharma ist... ► Artikel lesen | |
| EVOTEC | 5,095 | -3,96 % | EQS-NVR: Evotec SE: Veröffentlichung der Gesamtzahl der Stimmrechte nach § 41 WpHG mit dem Ziel der europaweiten Verbreitung | EQS Gesamtstimmrechtsmitteilung: Evotec SE
/ Veröffentlichung der Gesamtzahl der Stimmrechte
Evotec SE: Veröffentlichung der Gesamtzahl der Stimmrechte nach § 41 WpHG mit dem... ► Artikel lesen | |
| BB BIOTECH | 46,150 | -2,02 % | BB Biotech: "Wachstumsstarke Mid- und Small Caps bilden das Rückgrat unseres Portfolios": Mehr Einzelwerte und ... | ||
| MEDIGENE | 0,027 | -6,21 % | PTA-Adhoc: Medigene AG: Widerruf der Zulassung der Aktien der Medigene AG zum Handel im regulierten Markt | DJ PTA-Adhoc: Medigene AG: Widerruf der Zulassung der Aktien der Medigene AG zum Handel im regulierten Markt
Veröffentlichung von Insiderinformationen gemäß Artikel 17 MAR Schlagwort(e): Sonstiges... ► Artikel lesen | |
| QIAGEN | 30,705 | -0,36 % | DEUTSCHE BANK RESEARCH stuft QIAGEN NV auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat die Einstufung für Qiagen mit einem Kursziel von 43 US-Dollar auf "Buy" belassen. Das Interesse der Investoren habe vor allem dem Tuberkulosetest... ► Artikel lesen | |
| MODERNA | 38,365 | -3,13 % | Moderna, Inc.: Moderna to Present at Upcoming Conference in June 2026 | CAMBRIDGE, MA / ACCESS Newswire / June 2, 2026 / Moderna, Inc. (Nasdaq:MRNA), today announced its participation in the following upcoming investor conference:Goldman Sachs 47th Annual Global Healthcare... ► Artikel lesen | |
| PAION | 0,065 | -21,21 % | XFRA MISTRADE ANTRAG IN ISIN DE000A3E5EG5 WURDE STATTGEGEBEN | Dem Mistrade-Antrag wurde stattgegeben. Folgende Geschaefte in der ISIN DE000A3E5EG5, Wertpapier-Name: PAION AG INH O.N., wurden aufgehoben:ISIN Datum Zeit Volumen PreisDE000A3E5EG5 02.02.2026 08:13:40 300 0... ► Artikel lesen | |
| VALNEVA | 2,352 | -6,44 % | Original-Research: Valneva SE (von First Berlin Equity Research GmbH): Buy | Original-Research: Valneva SE - von First Berlin Equity Research GmbH
28.05.2026 / 17:08 CET/CEST
Veröffentlichung einer Research, übermittelt durch EQS News - ein Service der EQS Group.
Für den... ► Artikel lesen | |
| AMGEN | 280,90 | -0,72 % | Mesoblast: Während Amgen und Novo Nordisk den Biotech-Hype treiben, arbeitet dieser Zelltherapie-Player am operativen Durchbruch | ||
| EPIGENOMICS | 0,870 | 0,00 % | PTA-HV: Epigenomics AG: Einladung zur ordentlichen Hauptversammlung | DJ PTA-HV: Epigenomics AG: Einladung zur ordentlichen Hauptversammlung
Hauptversammlung gemäß -- 121 Abs. 4a AktG
Epigenomics AG: Einladung zur ordentlichen Hauptversammlung
Heidelberg... ► Artikel lesen | |
| NOVAVAX | 8,670 | -4,09 % | Novavax, Inc. (NVAX) Is a Trending Stock: Facts to Know Before Betting on It | ||
| STRYKER | 253,90 | -1,40 % | Stryker bringt das Pangea-Plattensystem auf den Markt und schließt den ersten Eingriff in Europa ab | Stryker (NYSE:SYK), ein weltweit führendes Unternehmen im Bereich Medizintechnik, gab heute die Markteinführung seines Pangea-Plattensystems in Europa bekannt, einer Plattensystem-Plattform zur Behandlung... ► Artikel lesen | |
| BIOGEN | 162,04 | -1,96 % | UCB and Biogen Announce Publication in The Lancet of Positive Dapirolizumab Pegol (DZP) Phase 3 Study Results in Systemic Lupus Erythematosus | Publication adds to previously reported findings from Phase 3 PHOENYCS GO study, demonstrating statistically significant improvement in disease activity at Week 48 with dapirolizumab pegol plus standard... ► Artikel lesen | |
| BIOFRONTERA | 2,510 | +2,87 % | PTA-News: Biofrontera AG: Biofrontera berichtet über die Ergebnisse des ersten Quartals 2026 | DJ PTA-News: Biofrontera AG: BIOFRONTERA BERICHTET ÜBER DIE ERGEBNISSE DES ERSTEN QUARTALS 2026
Unternehmensmitteilung für den Kapitalmarkt
Biofrontera AG: BIOFRONTERA BERICHTET ÜBER... ► Artikel lesen | |
| HEIDELBERG PHARMA | 2,680 | -2,19 % | EQS-HV: Heidelberg Pharma AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in Ladenburg mit dem Ziel der europaweiten Verbreitung gemäß §121 AktG | EQS-News: Heidelberg Pharma AG
/ Bekanntmachung der Einberufung zur Hauptversammlung
Heidelberg Pharma AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in... ► Artikel lesen |
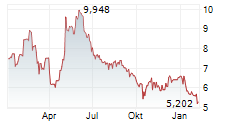
Das Unternehmen BIOCRYST PHARMACEUTICALS INC kann der Branche Biotechnologie zugeordnet werden. Die relative Kursveränderung beträgt aktuell -3,66 % bei einem Aktienkurs von 7,102 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu BIOCRYST PHARMACEUTICALS INC finden Sie auf Wallstreet Online.