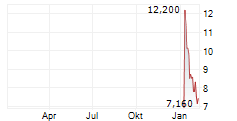- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 27.05. | TRANSTHERA-B (02617): NEXT DAY DISCLOSURE RETURN | - | HKEx | ||
| 27.05. | TRANSTHERA-B (02617): COMPLETION OF PLACING OF NEW H SHARES UNDER THE GENERAL MANDATE | - | HKEx | ||
| 26.05. | TRANSTHERA-B (02617): FORM OF PROXY FOR THE ANNUAL GENERAL MEETING TO BE HELD ON THURSDAY, 18 JUNE 2026 AT 10:30 A.M. | - | HKEx | ||
| 26.05. | TRANSTHERA-B (02617): NOTICE OF ANNUAL GENERAL MEETING | - | HKEx | ||
| 26.05. | TRANSTHERA-B (02617): CIRCULAR OF ANNUAL GENERAL MEETING | 1 | HKEx | ||
| 26.05. | TRANSTHERA-B (02617): VOLUNTARY ANNOUNCEMENT PHASE II CLINICAL TRIAL APPROVAL OF TINENGOTINIB IN COMBINATION WITH NOVEL HORMONE THERAPY FOR THE TREATMENT ... | - | HKEx | ||
| 26.05. | TRANSTHERA-B (02617): VOLUNTARY ANNOUNCEMENT TRANSTHERA SCIENCES (NANJING), INC. AND SHANGHAI ALLIST PHARMACEUTICALS CO., LTD. ANNOUNCE A COLLABORATION ... | 1 | HKEx | ||
| TRANSTHERA SCIENCES Aktie jetzt für 0€ handeln | |||||
| 22.05. | TRANSTHERA-B (02617): RULES RELATING TO THE 2026 H SHARE AWARD SCHEME | - | HKEx | ||
| 22.05. | TRANSTHERA-B (02617): FORM OF PROXY FOR THE EXTRAORDINARY GENERAL MEETING TO BE HELD ON 18 JUNE 2026 OR ANY ADJOURNMENT THEREOF | - | HKEx | ||
| 22.05. | TRANSTHERA-B (02617): NOTICE OF THE EXTRAORDINARY GENERAL MEETING | - | HKEx | ||
| 22.05. | TRANSTHERA-B (02617): (1) PROPOSED ADOPTION OF THE H SHARE AWARD SCHEME (2) PROPOSED AUTHORIZATION TO THE SCHEME ADMINISTRATOR TO DEAL WITH MATTERS RELATED ... | - | HKEx | ||
| 19.05. | TRANSTHERA-B Shares Down 8% on Placing at 18% Discount | 2 | AASTOCKS | ||
| 19.05. | TRANSTHERA-B Places Shares at 18% Discount w/HKD152M Net Proceeds | 1 | AASTOCKS | ||
| 19.05. | TRANSTHERA-B (02617): PLACING OF NEW H SHARES UNDER GENERAL MANDATE | 2 | HKEx | ||
| 18.05. | TRANSTHERA-B Announces Exploratory Clinical Results of Core Product Tiengetinib | 1 | AASTOCKS | ||
| 23.04. | TRANSTHERA-B (02617): ENVIRONMENTAL, SOCIAL AND GOVERNANCE REPORT 2025 | - | HKEx | ||
| 23.04. | TRANSTHERA-B (02617): ANNUAL REPORT 2025 | 3 | HKEx | ||
| 21.04. | TRANSTHERA-B (02617): NEXT DAY DISCLOSURE RETURN | 2 | HKEx | ||
| 21.04. | TRANSTHERA-B (02617): COMPLETION OF PLACING OF NEW H SHARES UNDER THE GENERAL MANDATE | - | HKEx | ||
| 21.04. | TRANSTHERA-B (02617): VOLUNTARY ANNOUNCEMENT TRANSTHERA SCIENCES (NANJING), INC. PRESENTED LINEAGE REPROGRAMMING AS A RESISTANCE MECHANISM TO ENDOCRINE ... | 1 | HKEx |
1 2 Weiter >>
38 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| MINERALYS THERAPEUTICS | 25,050 | -0,04 % | Mineralys Therapeutics, Inc.: Mineralys Therapeutics Announces Repurchase of Royalty Obligations in Tanabe License Agreement and Concurrent Financing | - Repurchase of lorundrostat royalty obligation in Tanabe license agreement for $200 million upfront and up to $100 million once certain commercial milestones are met - - $500 million committed... ► Artikel lesen | |
| PRAXIS PRECISION MEDICINES | 278,89 | -0,01 % | Praxis Precision Medicines, Inc.: Praxis Precision Medicines Provides Vormatrigine Program Update | POWER1 Study in highly refractory patients with focal onset seizures did not meet its primary success measure Secondary measure, the 50% response rate, was met and seizure reduction during the second... ► Artikel lesen | |
| QIAGEN | 30,975 | +0,88 % | DEUTSCHE BANK RESEARCH stuft QIAGEN NV auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat die Einstufung für Qiagen mit einem Kursziel von 43 US-Dollar auf "Buy" belassen. Das Interesse der Investoren habe vor allem dem Tuberkulosetest... ► Artikel lesen | |
| RECURSION PHARMACEUTICALS | 3,470 | 0,00 % | Recursion Pharmaceuticals: Recursion Reports Grant of Inducement Awards as Permitted by the Nasdaq Listing Rules | ||
| TANGO THERAPEUTICS | 21,470 | 0,00 % | Tango Therapeutics, Inc.: Tango Therapeutics Reports First Quarter 2026 Financial Results and Provides Business Highlights | First clinical data from PRMT5/RAS(ON) combination trial to be presented in 2026 Cash position of $380 million as of March 31, 2026, with runway into 2028 beyond anticipated key data inflection points... ► Artikel lesen | |
| ERASCA | 13,170 | -0,08 % | Erasca, Inc.: Erasca Reports First Quarter 2026 Business Updates and Financial Results | Robust monotherapy efficacy and generally well-tolerated safety results observed during dose escalation for ERAS-0015 in both KRAS G12X NSCLC and PDAC reinforce best-in-class potential across RAS-targeted... ► Artikel lesen | |
| COGENT BIOSCIENCES | 32,220 | -0,12 % | Cogent Biosciences, Inc.: Cogent Biosciences Announces Detailed Clinical Data from PEAK Phase 3 Trial with Bezuclastinib in Combination with Sunitinib in Gastrointestinal Stromal Tumors (GIST) at 2026 American Society of Clinical Oncology (ASCO) ... | Bezuclastinib combination is first treatment ever to demonstrate statistically significant advantage against active comparator in GIST patients; median PFS of 16.5 months versus 9.2 months (HR=0.50... ► Artikel lesen | |
| ENLIVEN THERAPEUTICS | 36,030 | 0,00 % | Enliven to host webcast on ELVN-001 trial data June 11 | ||
| IMMUNOVANT | 31,110 | -0,13 % | H.C. Wainwright Raises Price Target on Immunovant (IMVT) Following Positive Rheumatoid Arthritis Trial Data | ||
| SUMMIT THERAPEUTICS | 15,075 | +0,03 % | Cantor Fitzgerald reiterates Overweight on Summit Therapeutics stock | ||
| APOGEE THERAPEUTICS | 84,19 | 0,00 % | Weekly Buzz: AstraZeneca, JNJ Get FDA Nod; Eli Lilly To Acquire Curevo; APGE Advances Zumilokibart | NEW BRUNSWICK (dpa-AFX) - This week in the biotech space, there were regulatory approvals across the U.S. and Europe, collaborations, acquisitions, and positive data readouts spanning multiple... ► Artikel lesen | |
| STRUCTURE THERAPEUTICS | 37,500 | 0,00 % | Structure Therapeutics Inc.: Structure Therapeutics Appoints Matthew Lang, J.D. as Chief Operating Officer and General Counsel | SAN FRANCISCO, April 14, 2026 (GLOBE NEWSWIRE) -- Structure Therapeutics Inc. (NASDAQ: GPCR), a clinical-stage global biopharmaceutical company developing novel oral small molecule therapeutics for... ► Artikel lesen | |
| EDGEWISE THERAPEUTICS | 36,180 | -0,08 % | Servier to Acquire Edgewise Therapeutics' Muscular Dystrophy Business | Acquisition supports Servier's strategic ambition in rare neurology for patients with high unmet medical needs.Transaction includes sevasemten, a late-stage investigational treatment for... ► Artikel lesen | |
| RELAY THERAPEUTICS | 15,300 | 0,00 % | Relay Therapeutics Shares Jump 18% | ||
| CG ONCOLOGY | 54,40 | -0,04 % | CG Oncology Inc.: CG Oncology Reports Positive First Results from CORE-008 Cohort CX Phase 2 Trial Evaluating Intravesical Combination Therapy in High-Risk BCG-Exposed and BCG-Unresponsive Patients | - High CR rates at any time observed in the CIS-containing population with 85.7% and 92.3% in the ITT population and Efficacy Evaluable population, respectively - High-Grade - EFS in the overall intention-to-treat... ► Artikel lesen |
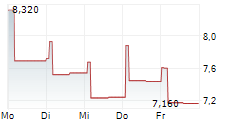
TRANSTHERA SCIENCES NANJING INC gehört der Branche Biotechnologie an. Die relative Kursveränderung beträgt aktuell -2,55 % bei einem Aktienkurs von 3,820 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu TRANSTHERA SCIENCES NANJING INC finden Sie auf Wallstreet Online.