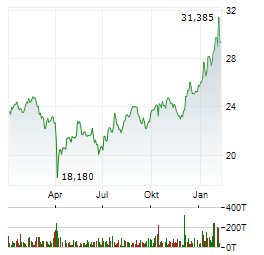
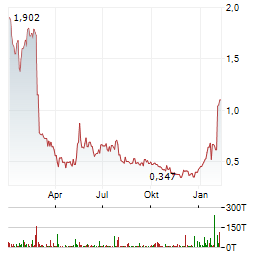
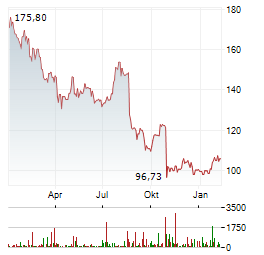
Chart-Typ:
Zeitraum:
| US-Silberimporte unter Druck: Prismo Metals rückt nahe Rio Tinto und BHP in den Fokus | Silber wird in den USA zum strategischen Engpass. Industrie, Solar, Elektronik und Verteidigung treiben den Bedarf, während die Importabhängigkeit hoch bleibt. Davon könnten heimische Projekte wie Silver... ► Artikel lesen |
| Welche Rohstoffe KI-Rechenzentren jetzt massiv benötigen - Kupfer-Aktien im Fokus | Anzeige / Werbung
Der weltweite Ausbau von KI-Infrastruktur entwickelt sich zunehmend zu einem neuen Treiber für den Rohstoffmarkt. Während viele Beobachtende vor allem auf Software, Chips oder Cloudanbieter
... ► Artikel lesen |
| MEGA-Multi-Metall-Fund...: Mogotes Metals: Discovery-Bohrung hebt Filo Sur auf ein neues Level! | Anzeige / WerbungMit dem 'Discovery-Hole', in dem eine hochgradige Multi-Metall-Mineralisierung entlang des Streichs der Filo-del-Sol-Lagerstätte getroffen wurde, könnte ein entscheidender Werttreiber... ► Artikel lesen |
| Dividendenbekanntmachungen (10.03.2026) | Unternehmen ISIN-Code Dividende (Währung) Dividende (EUR) ADAIRS LIMITED AU000000ADH2 0,055 AUD 0,0334 EUR ALCOA CORPORATION US0138721065 0,1 USD 0,0859 EUR AMEREN CORPORATION US0236081024 0... ► Artikel lesen |
| Angestellte Schweiz fordert Transparenz: CSL Behring schuldet Antworten und eine klare Standortstrategie | Olten (ots) - Angestellte Schweiz zeigt sich sehr besorgt über die Entwicklung bei CSL Behring: Mitarbeitende verdienen eine klare, transparente Strategie statt widersprüchlich wirkender Investitionsentscheide... ► Artikel lesen |
| Memo Therapeutics AG Enters into a Collaboration and Option Agreement with CSL for Development of Recombinant Polyclonal IgG Technology | PRESS RELEASE
Strategic research collaboration to advance MTx's recombinant polyclonal IgG technologyAgreement includes R&D funding for exploratory work, technology access and, assuming exercise... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu NEARMAP LTD finden Sie auf Wallstreet Online.