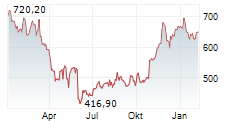
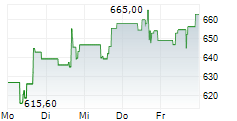
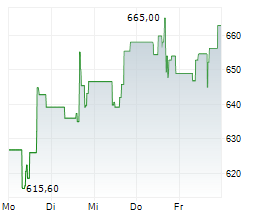
WASHINGTON (dpa-AFX) - Regeneron Pharmaceuticals said olatorepatide, an investigational obesity treatment it licensed from Hansoh Pharmaceutical Group, delivered positive Phase 3 trial results in Chinese patients with obesity or overweight.
The randomized, double-blind, placebo-controlled study enrolled 604 adults across 33 clinical sites in mainland China and evaluated once-weekly doses of 5 mg, 10 mg and 15 mg of olatorepatide over 48 weeks.
Results showed participants receiving the therapy achieved up to 19% average weight loss from baseline at week 48, while up to 97% of treated patients lost at least 5% of body weight, meeting the trial's co-primary endpoints.
'We are encouraged by the olatorepatide Phase 3 results in this Chinese population, which demonstrate not only meaningful weight loss but also a tolerability profile that could make a real difference in patients' day-to-day experience on treatment,' said George D. Yancopoulos, M.D., Ph.D., Board co-Chair, President, and Chief Scientific Officer at Regeneron. 'We are on track to advance into our registrational program later this year and are eager to see the results from our pivotal trials.'
Olatorepatide, a dual GLP-1/GIP receptor agonist, also demonstrated a favorable gastrointestinal tolerability profile. Reported rates of nausea were below 10%, while vomiting incidence was below 5%, with relatively low treatment discontinuation.
Under the companies' agreement, Hansoh retains development and commercialization rights in Greater China, while Regeneron holds exclusive development and commercial rights outside mainland China, Hong Kong and Macau.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News