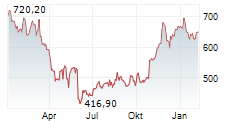
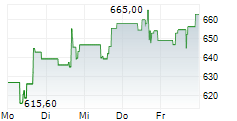
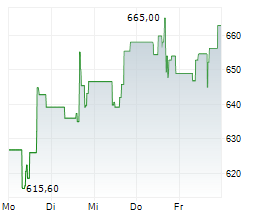
WASHINGTON (dpa-AFX) - Regeneron Pharmaceuticals (REGN) reported positive Phase 3 results for cemdisiran in Generalized Myasthenia Gravis, with the NIMBLE trial meeting primary and key secondary endpoints at 24 weeks.
Cemdisiran-treated patients showed significant improvements in MG-ADL and QMG scores versus placebo, with placebo-adjusted gains of 2.3 points and 2.8 points, respectively.
Clinical benefits appeared within two weeks and were sustained through the study period. Notably, 76.6% of patients achieved ?3-point MG-ADL improvement compared to 44.1% on placebo.
The investigational siRNA therapy targets complement factor C5 and is administered once every 12 weeks, offering a potentially more convenient treatment option.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News