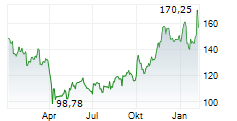
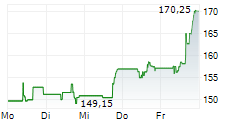
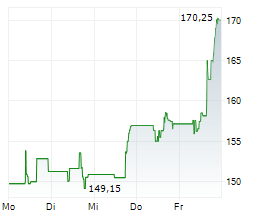
WESTON (dpa-AFX) - Eisai Co. Ltd (ESALY.PK) and Biogen Inc. (BIIB) on Friday said the U.S. Food and Drug Administration has extended by three months the review of LEQEMBI IQLIK as a starting dose for the treatment of early Alzheimer's disease.
The new Prescription Drug User Fee Act (PDUFA) action date is August 24, 2026.
The FDA requested additional information during the review process and has not raised any concerns about the drug's approvability as a starting dose, the companies said.
LEQEMBI has been approved in more than 50 countries for the treatment of early Alzheimer's disease.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News