- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 01.05. | Baird initiates Lyell Immunopharma stock with outperform rating | 1 | Investing.com | ||
| 01.05. | BofA reiterates Lyell Immunopharma stock Underperform on rival exit | 3 | Investing.com | ||
| 23.03. | H.C. Wainwright reiterates Lyell Immunopharma stock rating at buy | 3 | Investing.com | ||
| 16.03. | Lyell Immunopharma: Citizens bestätigt "Market Outperform" nach Fortschritten in Studien | 1 | Investing.com Deutsch | ||
| 16.03. | Citizens reiterates Lyell Immunopharma stock rating on trial progress | 1 | Investing.com | ||
| LYELL IMMUNOPHARMA Aktie jetzt für 0€ handeln | |||||
| 12.03. | Lyell Immunopharma, Inc. - S-8, Securities to be offered to employees in employee benefit plans | 1 | SEC Filings | ||
| 12.03. | Lyell Immunopharma, Inc. - 10-K, Annual Report | 1 | SEC Filings | ||
| 09.03. | Lyell Immunopharma closes $50M equity tranche, names new CFO | 1 | Investing.com | ||
| 09.03. | Lyell Immunopharma, Inc: Lyell Immunopharma Announces Closing of Additional $50 Million Tranche of Equity Private Placement and Appointment of Smital Shah as Chief Financial and Business Officer | 1 | GlobeNewswire (USA) | ||
| 09.03. | Lyell Immunopharma, Inc. - 8-K, Current Report | 1 | SEC Filings | ||
| 09.03. | Citizens initiates Lyell Immunopharma stock at outperform, $34 target | 1 | Investing.com | ||
| 12.02. | Lyell Immunopharma, Inc: Lyell Immunopharma Announces Initiation of Patient Dosing in First-of-Its-Kind Phase 3 Head-To-Head CAR T-Cell Clinical Trial in Aggressive Large B-Cell Lymphoma | 1.098 | GlobeNewswire (Europe) | PiNACLE - H2H will evaluate the efficacy and safety of rondecabtagene autoleucel (ronde-cel) versus Investigator's choice of approved CD19 CAR T-cell therapies in patients with aggressive large B-cell... ► Artikel lesen | |
| 22.12.25 | Lyell Immunopharma pops on recent ASH data, immunotherapy expansion | 4 | Seeking Alpha | ||
| 07.12.25 | Lyell Immunopharma, Inc: Lyell Immunopharma Presents New Clinical Data from Ongoing Trial of Ronde-Cel Showing High Rates of Durable Complete Responses in Patients with Large B-cell Lymphoma at the 67th ASH Annual Meeting and Exposition | 2 | GlobeNewswire (USA) | ||
| 05.12.25 | Lyell Immunopharma, Inc. - 8-K, Current Report | - | SEC Filings | ||
| 10.11.25 | Lyell Immunopharma, Inc: Lyell Immunopharma Acquires Exclusive Global Rights to a Next-Generation CAR T-Cell Product Candidate in Clinical Development for Metastatic Colorectal Cancer | 215 | GlobeNewswire (Europe) | LYL273 has demonstrated a 67% overall response rate, an 83% disease control rate, and a manageable safety profile at the highest dose level studied to date in patients with refractory metastatic colorectal... ► Artikel lesen | |
| 12.08.25 | Lyell Immunopharma, Inc: Lyell Immunopharma Reports Business Highlights and Financial Results for the Second Quarter 2025 | 444 | GlobeNewswire (Europe) | Presented positive new clinical data demonstrating high rates of durable complete responses from the Phase 1/2 trial of LYL314 for the treatment of aggressive large B-cell lymphomaInitiated the PiNACLE... ► Artikel lesen | |
| 17.06.25 | Lyell Immunopharma, Inc: Lyell Immunopharma Announces Positive New Clinical Data Demonstrating High Rates of Durable Complete Responses from the Phase 1/2 Trial of LYL314 for the Treatment of Aggressive Large B-cell Lymphoma | 267 | GlobeNewswire (Europe) | LYL314 demonstrated robust clinical responses, with an 88% overall response rate and a 72% complete response rate in patients treated in the third- or later-line setting (N = 25)71% of patients with... ► Artikel lesen | |
| 09.06.25 | Lyell Immunopharma, Inc: Lyell Immunopharma Strengthens Clinical and Commercial Capabilities with Key Board and Executive Appointments | 324 | GlobeNewswire (Europe) | Mark J. Bachleda, PharmD, MBA appointed as independent member of the Board of DirectorsDavid Shook, MD appointed as Chief Medical Officer, Mark Meltz, JD as General Counsel and Corporate Secretary... ► Artikel lesen |
19 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| MEDIGENE | 0,037 | +18,59 % | MEDIGENE AG im Fokus - ruhige Einordnung der Schwäche | ||
| AMGEN | 292,50 | +0,05 % | Endokrine Orbitopathie: Amgen lenkt den Blick auf Versorgungslücken und gesellschaftliche Folgen | München (ots) - Bis zu zwei Jahre Diagnose-Wartezeit und Beeinträchtigung von Lebensqualität, Beruf und sozialer Teilhabe[1-4]Zum Weltschilddrüsentag am 25. Mai 2026 rückt das Biotechnologieunternehmen... ► Artikel lesen | |
| BIOGEN | 166,64 | -0,22 % | XETR DELETION OF INSTRUMENTS FROM XETRA - 22.05.2026 | The following instruments on XETRA do have their last trading day on 22.05.2026.Die folgenden Instrumente auf XETRA haben ihren letzten Handelstag am 22.05.2026..ISIN Name US09062X1037 Biogen Inc.US2521311074 DexCom... ► Artikel lesen | |
| EDITAS MEDICINE | 2,310 | -2,94 % | Editas Medicine, Inc.: Editas Medicine Reports New Preclinical Data Demonstrating Progress of EDIT-401 as Potential Treatment for Hyperlipidemia at the American Society of Gene and Cell Therapy 2026 Annual Meeting | CAMBRIDGE, Mass., May 14, 2026 (GLOBE NEWSWIRE) -- Editas Medicine, Inc. (Nasdaq: EDIT), a pioneering gene editing company developing transformative medicines for serious diseases, shared new preclinical... ► Artikel lesen | |
| LAKEFRONT BIOTHERAPEUTICS | 23,920 | +0,67 % | Galapagos NV: Galapagos Reports First Quarter 2026 Financial Results and Provides Business Update | Galapagos' name change to Lakefront Biotherapeutics approved at the Company's EGM (Euronext & NASDAQ: LKFT)Collaboration Agreement with Gilead to advance potential First and Best-in-Class T Cell Engager... ► Artikel lesen | |
| IMMATICS | 10,160 | +1,50 % | Immatics Announces First Quarter 2026 Financial Results and Business Update | SUPRAME Phase 3 interim and final analysis for PRAME cell therapy, anzu-cel, expected to be triggered in 2026, advancing toward the Company's first commercial launch planned in 2027Multiple key clinical... ► Artikel lesen | |
| AUTOLUS THERAPEUTICS | 1,510 | +2,72 % | Autolus Therapeutics plc: Autolus Therapeutics to Present Clinical Data Update at the American Society of Clinical Oncology Annual Meeting 2026 | ||
| TANGO THERAPEUTICS | 20,240 | 0,00 % | Tango Therapeutics, Inc.: Tango Therapeutics Reports First Quarter 2026 Financial Results and Provides Business Highlights | First clinical data from PRMT5/RAS(ON) combination trial to be presented in 2026 Cash position of $380 million as of March 31, 2026, with runway into 2028 beyond anticipated key data inflection points... ► Artikel lesen | |
| TSCAN THERAPEUTICS | 1,060 | 0,00 % | TScan Therapeutics, Inc. - 8-K, Current Report | ||
| SUMMIT THERAPEUTICS | 16,620 | 0,00 % | This Summit Therapeutics Analyst Is No Longer Bullish; Here Are Top 2 Downgrades For Friday | ||
| CG ONCOLOGY | 64,25 | 0,00 % | Wolfe Research startet Coverage für CG Oncology mit "Peerperform" | ||
| QIAGEN | 30,290 | +0,65 % | QIAGEN N.V.: QIAGEN to advance AI-driven drug discovery with graph-based AI and curated bioinformatics knowledge with NVIDIA | Collaboration integrates NVIDIA accelerated computing and the NVIDIA BioNeMo platform with QIAGEN Digital Insights' curated biomedical knowledge bases Goal to advance how disease mechanisms... ► Artikel lesen | |
| PRAXIS PRECISION MEDICINES | 343,26 | -0,09 % | Praxis Precision Medicines, Inc.: Praxis Precision Medicines Provides Corporate Update and Reports First Quarter 2026 Financial Results | FDA accepted the new drug application (NDA) for ulixacaltamide in Essential Tremor with a PDUFA target action date of January 29, 2027, and the NDA for relutrigine, with priority review, in SCN2A... ► Artikel lesen | |
| COGENT BIOSCIENCES | 32,640 | -0,09 % | Cogent Biosciences: Aktie legt vor wichtigen Studiendaten um 4 % zu | ||
| GEOVAX LABS | 3,630 | -0,27 % | EQS-News: GeoVax, Inc.: GeoVax Comments on Escalating Bundibugyo Ebola Outbreak and Growing Need for Flexible Biodefense Vaccine Platforms | EQS-News: GeoVax, Inc.
/ Key word(s): Science
GeoVax Comments on Escalating Bundibugyo Ebola Outbreak and Growing Need for Flexible Biodefense Vaccine Platforms 20.05.2026 / 16:06... ► Artikel lesen |
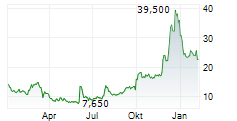
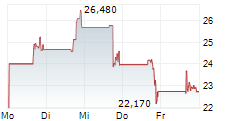
LYELL IMMUNOPHARMA INC gehört der Branche Biotechnologie an. Der aktuelle Kurs liegt bei 17,450 Euro und damit -0,06 % im Minus.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu LYELL IMMUNOPHARMA INC finden Sie auf Wallstreet Online.