- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 24.04. | NeuroSense Therapeutics Ltd. - 6-K, Report of foreign issuer | - | SEC Filings | ||
| 03.04. | NeuroSense Therapeutics receives Nasdaq notices over listing requirements | 20 | Seeking Alpha | ||
| 03.04. | NeuroSense Therapeutics Announces Receipt of Nasdaq Notifications Regarding Minimum Bid Price and Market Value Requirements | 239 | PR Newswire | Notifications have no immediate effect on the listing or trading of the Company's securities on Nasdaq
Nasdaq has provided the Company until September 29, 2026... ► Artikel lesen | |
| 31.03. | NeuroSense Therapeutics reports FY results, GAAP EPS of $0.44 | 2 | Seeking Alpha | ||
| 31.03. | NeuroSense Therapeutics Reports Year End 2025 Financial Results and Provides Business Update | 371 | PR Newswire | 2025 marked transition of PrimeC into a late-stage clinical asset with FDA-cleared Phase 3 program in ALS
Statistically significant survival benefit demonstrated... ► Artikel lesen | |
| NEUROSENSE THERAPEUTICS Aktie jetzt für 0€ handeln | |||||
| 31.03. | NeuroSense Therapeutics Ltd. - 6-K, Report of foreign issuer | 1 | SEC Filings | ||
| 31.03. | NeuroSense Therapeutics Ltd. - 20-F, Annual and transition report of foreign private issuers | 2 | SEC Filings | ||
| 24.03. | NeuroSense Therapeutics Ltd. - 6-K, Report of foreign issuer | 1 | SEC Filings | ||
| 16.03. | NeuroSense: JAMA Neurology Publishes Results from PARADIGM Phase 2b Trial of PrimeC in ALS Demonstrating Meaningful Clinical Outcomes and Biological Activity | 270 | PR Newswire | The Journal of the American Medical Association (JAMA Neurology) publication highlights consistent clinical and biomarker findings from NeuroSense PARADIGM Phase... ► Artikel lesen | |
| 11.03. | NeuroSense Therapeutics Ltd. - 6-K, Report of foreign issuer | 6 | SEC Filings | ||
| 10.03. | Roth/MKM initiates NeuroSense stock with buy on ALS drug potential | 5 | Investing.com | ||
| 18.02. | NeuroSense Therapeutics Ltd. - 6-K, Report of foreign issuer | 3 | SEC Filings | ||
| 09.02. | Neurosense-Aktie legt nach Patent-Erfolg in Australien zu | 2 | Investing.com Deutsch | ||
| 09.02. | NeuroSense Therapeutics Ltd. - 6-K, Report of foreign issuer | 2 | SEC Filings | ||
| 21.01. | NeuroSense granted U.S. patent for treatment of Alzheimer's disease | 9 | Seeking Alpha | ||
| 06.01. | NeuroSense Therapeutics Ltd. - 6-K, Report of foreign issuer | - | SEC Filings | ||
| 22.12.25 | NeuroSense Therapeutics Ltd. - 6-K, Report of foreign issuer | - | SEC Filings | ||
| 22.12.25 | NeuroSense Confirms Favorable Safety and Tolerability of PrimeC in an Alzheimer's Phase 2 Study | 615 | PR Newswire | Clinical and Biomarker Outcomes Expected to Be Reported in Q1 2026
CAMBRIDGE, Mass., Dec. 22, 2025 /PRNewswire/ -- NeuroSense Therapeutics Ltd. (NASDAQ: NRSN) ("NeuroSense")... ► Artikel lesen | |
| 04.12.25 | NeuroSense Therapeutics Ltd. - 6-K, Report of foreign issuer | 1 | SEC Filings | ||
| 24.11.25 | NeuroSense Receives FDA Clearance to Initiate Pivotal Phase 3 Trial for PrimeC in ALS | 470 | PR Newswire | CAMBRIDGE, Mass., Nov. 24, 2025 /PRNewswire/ -- NeuroSense Therapeutics Ltd. (Nasdaq: NRSN) ("NeuroSense"), a late-clinical stage biotechnology company developing... ► Artikel lesen |
1 2 Weiter >>
21 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| EVOTEC | 5,160 | +1,57 % | Shortseller-Positionen aktuell: u.a. Evotec, FMC, Gerresheimer, Hypoport, Nagarro, Nordex, Suss MicroTec | Wer Aktien leerverkauft, also auf fallende Kurse spekuliert (Shortselling), unterliegt bestimmten Transparenzpflichten. Aktuelle Meldungen über Leerverkäufe geben Aufschluss darüber. Diese Vorgaben... ► Artikel lesen | |
| MODERNA | 40,995 | +0,77 % | Moderna Flu Shot Heads To FDA Advisory Panel | ||
| VALNEVA | 2,492 | -0,20 % | Valneva Aktie: Prognose kassiert - die Börse schaut woanders hin | Valneva hat das erste Quartal 2026 mit einem deutlichen Umsatzrückgang abgeschlossen und die Jahresprognose gesenkt. Der Aktienkurs reagierte nicht entscheidend, denn der Markt richtet seinen Blick... ► Artikel lesen | |
| PALATIN TECHNOLOGIES | 14,380 | 0,00 % | Palatin Technologies, Inc.: Palatin to Begin Trading on Nasdaq Stock Exchange | PRINCETON, N.J., May 18, 2026 /PRNewswire/ -- Palatin Technologies, Inc. (NYSE American: PTN), a biopharmaceutical company focused on the discovery and development... ► Artikel lesen | |
| VIVOSIM LABS | 1,340 | 0,00 % | VivoSim Labs, Inc.: VivoSim Appoints Arumugham (Ragoo) Raghunathan as Vice President of Global Sales | SAN DIEGO, March 03, 2026 (GLOBE NEWSWIRE) -- VivoSim Labs, Inc. (Nasdaq: VIVS) (the "Company" or "VivoSim"), a provider of next-generation New Approach Methodologies (NAMs) for preclinical safety... ► Artikel lesen | |
| SANGAMO THERAPEUTICS | 0,126 | -100,00 % | Sangamo anticipates completing ST-920 BLA submission as early as this summer while appealing NASDAQ delisting in June | ||
| INTELLIA THERAPEUTICS | 10,600 | -0,56 % | Can Intellia's Pipeline Push Drive Long-Term Growth Amid Rivalry? | ||
| CARDIOL THERAPEUTICS | 1,126 | -1,23 % | Original-Research: Cardiol Therapeutics Inc (von First Berlin Equity Research GmbH): BUY | Original-Research: Cardiol Therapeutics Inc - from First Berlin Equity Research GmbH
30.04.2026 / 13:07 CET/CEST
Dissemination of a Research, transmitted by EQS News - a service of EQS Group.
The... ► Artikel lesen | |
| ARBUTUS BIOPHARMA | 3,750 | -1,83 % | Arbutus Biopharma Corporation: Arbutus Reports First Quarter 2026 Financial Results and Provides Corporate Update | Q1 total revenue of $179.1M includes estimated license revenue from Genevant related to litigation settlement with Moderna Strong financial position with cash, cash equivalents and marketable securities... ► Artikel lesen | |
| XOMA ROYALTY | 35,600 | -1,11 % | Ligand Pharmaceuticals und XOMA Royalty passen Fusionsvereinbarung an | ||
| CELLECTAR BIOSCIENCES | 3,170 | 0,00 % | Cellectar Biosciences, Inc. - S-1, General form for registration of securities | ||
| AIM IMMUNOTECH | 0,073 | -100,00 % | AIM ImmunoTech Inc.: AIM ImmunoTech Announces $2.4 Million Financing, Including Concurrent Registered Direct Offering and Private Placement | OCALA, Fla., May 20, 2026 (GLOBE NEWSWIRE) -- AIM ImmunoTech Inc. (NYSE American: AIM) ("AIM" or the "Company", today announced that it has entered into definitive agreements for a registered direct... ► Artikel lesen | |
| NOVOCURE | 14,710 | -4,54 % | Novocure Reports First Quarter 2026 Financial Results | Quarterly net revenues of $174 million, up 12% year-over-year, driven by global growth in Optune Gio
Optune Pax approved by the U.S. FDA and launched for the treatment of locally advanced pancreatic... ► Artikel lesen | |
| PHARMING | 1,120 | -0,88 % | Pharming Group N.V.: Pharming Group reports first quarter 2026 financial results; on track for Joenja U.S. pediatric label expansion and launches in Japan and Europe in 2026 | First quarter 2026 total revenues were US$72.4 million, an 8% decrease compared to the first quarter 2025RUCONEST revenue was US$58.4 million, a 15% decrease compared to the first quarter 2025, mainly... ► Artikel lesen | |
| TRAWS PHARMA | 1,500 | +0,67 % | Traws Pharma, Inc.: Traws Pharma Provides Business Highlights and Reports Q1 2026 Financial Results | Private financing of up to $60M offering of common stock (with $10M upfront) and milestone-based warrants expected to support operations into Q1 2027 Tivoxavir marboxil advancing towards a human influenza... ► Artikel lesen |
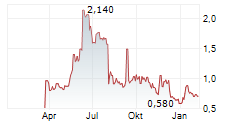
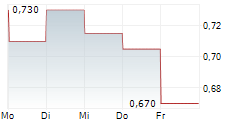
NEUROSENSE THERAPEUTICS LTD gehört der Branche Biotechnologie an. Die relative Kursveränderung beträgt aktuell -4,65 % bei einem Aktienkurs von 0,615 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu NEUROSENSE THERAPEUTICS LTD finden Sie auf Wallstreet Online.