- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| Di | Citizens reiterates Rapport Therapeutics stock rating on competitor trial data | 2 | Investing.com | ||
| 18.05. | Rapport Therapeutics: Truist nimmt Beobachtung mit Kaufempfehlung auf und hebt Kursziel an | 2 | Investing.com Deutsch | ||
| 18.05. | Truist assumes Rapport Therapeutics stock with buy on epilepsy drug | 2 | Investing.com | ||
| 07.05. | Raymond James reiterates Strong Buy on Rapport Therapeutics stock | 3 | Investing.com | ||
| 07.05. | Rapport Therapeutics GAAP EPS of -$0.42, revenue of $0.02M | 4 | Seeking Alpha | ||
| 07.05. | Rapport Therapeutics, Inc. - 10-Q, Quarterly Report | - | SEC Filings | ||
| RAPPORT THERAPEUTICS Aktie jetzt für 0€ handeln | |||||
| 07.05. | Rapport Therapeutics, Inc. - 8-K, Current Report | - | SEC Filings | ||
| 21.04. | Rapport Therapeutics reports extended efficacy data for RAP-219 | 3 | Investing.com | ||
| 21.04. | Rapport Therapeutics, Inc.: Rapport Therapeutics Presents RAP-219 Focal Onset Seizure Phase 2a Follow-up Period Results Demonstrating Sustained Seizure Reduction at the 2026 American Academy of Neurology Annual Meeting | 2 | GlobeNewswire (USA) | ||
| 21.04. | Rapport Therapeutics, Inc. - 8-K, Current Report | - | SEC Filings | ||
| 01.04. | Rapport Therapeutics, Inc.: Rapport Therapeutics to Present New Phase 2 Treatment Follow-Up Data for RAP-219 in Focal Onset Seizures at the 2026 American Academy of Neurology Annual Meeting | 3 | GlobeNewswire (USA) | ||
| 16.03. | Truist reiterates Rapport Therapeutics stock rating on phase 3 plans | 4 | Investing.com | ||
| 16.03. | Rapport Therapeutics: Truist bestätigt Kaufempfehlung nach beschleunigten Phase-3-Plänen | 11 | Investing.com Deutsch | ||
| 11.03. | Stifel reiterates Rapport Therapeutics stock rating after results | 5 | Investing.com | ||
| 11.03. | Jones Trading reiterates Rapport Therapeutics stock rating on epilepsy progress | 4 | Investing.com | ||
| 11.03. | BTIG raises Rapport Therapeutics stock price target on trial progress | 2 | Investing.com | ||
| 10.03. | Rapport Therapeutics GAAP EPS of -$0.72 misses by $0.03 | 1 | Seeking Alpha | ||
| 10.03. | Rapport Therapeutics, Inc. - S-8, Securities to be offered to employees in employee benefit plans | 1 | SEC Filings | ||
| 10.03. | Rapport Therapeutics, Inc. - 10-K, Annual Report | 1 | SEC Filings | ||
| 10.03. | Rapport Therapeutics, Inc. - 8-K, Current Report | 3 | SEC Filings |
1 2 Weiter >>
36 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| EVOTEC | 5,115 | +1,79 % | EQS-NVR: Evotec SE: Veröffentlichung der Gesamtzahl der Stimmrechte nach § 41 WpHG mit dem Ziel der europaweiten Verbreitung | EQS Gesamtstimmrechtsmitteilung: Evotec SE
/ Veröffentlichung der Gesamtzahl der Stimmrechte
Evotec SE: Veröffentlichung der Gesamtzahl der Stimmrechte nach § 41 WpHG mit dem... ► Artikel lesen | |
| QIAGEN | 32,630 | +5,34 % | DEUTSCHE BANK RESEARCH stuft QIAGEN NV auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat die Einstufung für Qiagen mit einem Kursziel von 43 US-Dollar auf "Buy" belassen. Das Interesse der Investoren habe vor allem dem Tuberkulosetest... ► Artikel lesen | |
| CRISPR THERAPEUTICS | 49,295 | +9,73 % | CRISPR Therapeutics Stock Is Absurdly Cheap -- Here's Why Analysts See 437% Upside Potential | ||
| VIKING THERAPEUTICS | 25,950 | +2,57 % | XETR NEW INSTRUMENTS AVAILABLE ON XETRA - 04.06.2026 | Folgende Wertpapiere werden in den Handel an folgenden Handelsplätzen (MIC Code) im Quotation Board an der Frankfurter Wertpapierbörse (FWB) aufgenommen. Die Notierung an anderen Handelsplätzen im Quotation... ► Artikel lesen | |
| EDITAS MEDICINE | 2,660 | +10,83 % | Editas Medicine, Inc.: Editas Medicine Reports New Preclinical Data Demonstrating Progress of EDIT-401 as Potential Treatment for Hyperlipidemia at the American Society of Gene and Cell Therapy 2026 Annual Meeting | CAMBRIDGE, Mass., May 14, 2026 (GLOBE NEWSWIRE) -- Editas Medicine, Inc. (Nasdaq: EDIT), a pioneering gene editing company developing transformative medicines for serious diseases, shared new preclinical... ► Artikel lesen | |
| BIOCRYST PHARMACEUTICALS | 7,230 | +1,57 % | BioCryst Ireland, a Neopharmed Gentili company: Hereditary angioedema, a European initiative to shine a spotlight on a still under-recognized condition | Ahead of World HAE Day, BioCryst Ireland, a Neopharmed Gentili company, organised an international event in Milan bringing together leading experts from across Europe to raise awareness of... ► Artikel lesen | |
| BIOMARIN PHARMACEUTICAL | 48,530 | +2,93 % | BioMarin Pharmaceutical Inc. (BMRN) A Stock that Could 10x by 2030 on Pipeline Development | ||
| ABIVAX | 88,95 | +16,05 % | Biotech Hoffnung erhält Dämpfer: Abivax Aktie stürzt nach Höhenflug ab Krebs Signale belasten Investmentstory | Die Abivax Aktie hat nach ihrem starken Höhenflug einen herben Rückschlag erlitten. Auslöser sind neue Sicherheitsfragen rund um den Hoffnungsträger Obefazimod. Obwohl der Wirkstoff in klinischen Studien... ► Artikel lesen | |
| EXELIXIS | 45,080 | +1,31 % | Exelixis präsentiert positive Studiendaten zu Cabometyx bei neuroendokrinen Tumoren | ||
| SAREPTA THERAPEUTICS | 13,880 | -0,86 % | Biotech-Player: Zelltherapie als Comeback-Story im Milliardenmarkt. Neue Ideen nach Corona von BioNTech, Qiagen und Biotech AG | ||
| HALOZYME THERAPEUTICS | 61,98 | +2,96 % | Benchmark reiterates Halozyme stock Buy rating on long-term potential | ||
| PACIFIC BIOSCIENCES OF CALIFORNIA | 1,403 | +2,82 % | Here's Why You Should Add PacBio Stock to Your Portfolio Now | ||
| BASILEA | 54,10 | -0,18 % | Basilea Pharmaceutica International Ltd, Allschwil: Basilea erhält weitere USD 13 Mio. von BARDA zur Entwicklung des neuartigen Antibiotikums Ceftibuten-Ledaborbactam | Ad hoc-Mitteilung gemäss Art. 53 KR
Allschwil, 28. Mai 2026
Basilea Pharmaceutica AG, Allschwil (SIX: BSLN), ein biopharmazeutisches Unternehmen mit bereits vermarkteten Produkten und dem Ziel... ► Artikel lesen | |
| IMMUNITYBIO | 6,340 | +2,26 % | ImmunityBio presents ANKTIVA data at ASCO meeting | ||
| MICROBOT MEDICAL | 1,710 | +0,88 % | Lucid Capital Markets initiates Microbot Medical stock with buy rating |
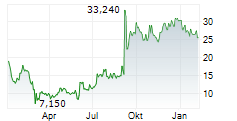
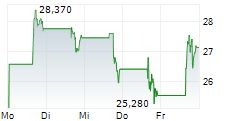
RAPPORT THERAPEUTICS INC gehört der Branche Biotechnologie an. Mit einer aktuellen Kursveränderung von 0,000 (0,00 %) liegt der Kurs bei 37,490 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu RAPPORT THERAPEUTICS INC finden Sie auf Wallstreet Online.