PARIS (dpa-AFX) - Regeneron Pharmaceuticals, Inc. (REGN) and Sanofi SA (SNY) Friday said the European Medicines Agencys Committee for Medicinal Products for Human Use (CHMP) has issued a positive opinion for Dupixent to treat chronic spontaneous urticaria (CSU) in children aged 2 to 11 years.
CSU is a chronic, inflammatory skin disease driven in part by type 2 inflammation.
The recommendation is supported by data from the LIBERTY-CUPID clinical trial program, including two Phase 3 trials, Study A and Study C, involving children aged 6 to 11 years, as well as the single-arm Phase 3 CUPIDKids trial in children aged 2 to 11 years.
Dupixent is already approved for CSU in certain adults and adolescents in several jurisdictions, including the United States, the European Union and Japan. In the U.S., the therapy is under review for certain children aged 2 to 11 years, with a decision expected by April 2026.
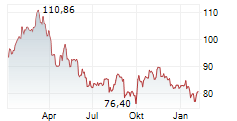
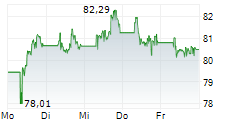
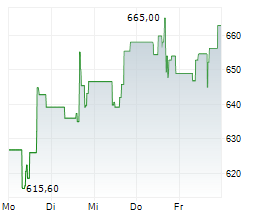
On Thursday, Regeneron shares closed at $770.79, down 1.72%, while Sanofi shares rose 1.6% to $48.35.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News