WASHINGTON (dpa-AFX) - Formycon AG (FYB.DE, FYBGF), an independent German biotechnology company, on Thursday announced that it entered into a settlement and license agreement with Regeneron Pharmaceuticals, Inc. (REGN) and Bayer Aktiengesellschaft (BAYN.DE, BAYRY).
The agreement secures a May 2026 launch date for its aflibercept 2 mg biosimilar FYB203 in Europe and other key markets and resolves all related patent disputes.
Market launches by commercialization partners across Europe are scheduled to begin in May 2026.
The agreement concluded alongside license partner Klinge Biopharma GmbH, covers Europe as well as key markets in Latin America and the Asia-Pacific region.
The settlement fully resolves all pending patent litigations related to the EU-approved Eylea 2 mg biosimilars AHZANTIVE and Baiama.
The company had previously secured a U.S. license date for FYB203 in the fourth quarter of 2026 under a separate agreement with Regeneron.
AHZANTIVE and Baiama have been approved by the European Medicines Agency for the treatment of neovascular age-related macular degeneration and other serious retinal diseases.
The biosimilars contain aflibercept, which inhibits vascular endothelial growth factor, a driver of abnormal blood vessel growth in the retina that can lead to severe vision impairment.
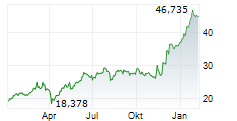
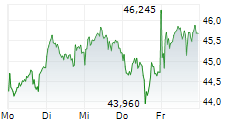
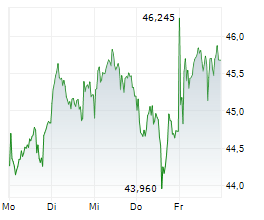
On Wednesday, FYB.DE closed trading 0.10% lesser at EUR 19.82 on the XETRA.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News