- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 26.05. | Cantor Fitzgerald reiterates Artiva Biotherapeutics stock rating | 2 | Investing.com | ||
| 26.05. | Cantor Fitzgerald bekräftigt "Overweight"-Rating für Artiva Biotherapeutics | 4 | Investing.com Deutsch | ||
| 19.05. | Artiva to present AlloNK therapy data at EULAR congress | 1 | Investing.com | ||
| 19.05. | Artiva Biotherapeutics, Inc.: Artiva Biotherapeutics Announces Multiple AlloNK Data Presentations at EULAR 2026 Congress, Including Late-Breaking Oral Presentation Highlighting Clinical Efficacy Comparable to Autologous CAR T-Cell Therapy in ... | 5 | GlobeNewswire (USA) | ||
| 19.05. | Artiva Biotherapeutics appoints Diego Miralles as president, R&D head | 1 | Investing.com | ||
| 19.05. | Artiva Biotherapeutics beruft Diego Miralles zum President und Leiter für Forschung und Entwicklung | 1 | Investing.com Deutsch | ||
| 19.05. | Artiva Biotherapeutics, Inc. - 8-K, Current Report | 1 | SEC Filings | ||
| ARTIVA BIOTHERAPEUTICS Aktie jetzt für 0€ handeln | |||||
| 19.05. | Artiva Biotherapeutics, Inc.: Artiva Biotherapeutics Appoints Veteran Biotech Executive and Drug Developer Diego Miralles, M.D., as President and Head of Research and Development | 778 | GlobeNewswire (Europe) | Veteran biotechnology executive with more than 20 years of leadership experience spanning research, clinical development, commercialization and company building Proven track record advancing multiple... ► Artikel lesen | |
| 11.05. | H.C. Wainwright raises Artiva Biotherapeutics price target to $35 | 2 | Investing.com | ||
| 11.05. | GC Cell's Artiva Raises $300 Million for Phase 3 RA Trial | 2 | BusinessKorea | ||
| 08.05. | Artiva Biotherapeutics Stock Slides Despite Positive Trial Data | 2 | Benzinga.com | ||
| 08.05. | Artiva rises after mid-stage trial data for lead asset in rheumatoid arthritis | 1 | Seeking Alpha | ||
| 08.05. | Artiva BioTherapeutics GAAP EPS of -$0.95 | 1 | Seeking Alpha | ||
| 08.05. | Artiva Biotherapeutics Prices $300 Mln Stock, Pre-Funded Warrants Offering; Shares Rise | 1 | RTTNews | ||
| 08.05. | Artiva reports clinical trial data for rheumatoid arthritis therapy | 3 | Investing.com | ||
| 08.05. | Artiva Biotherapeutics, Inc.: Artiva Biotherapeutics Reports First Quarter 2026 Financial Results and Recent Business Highlights | 247 | GlobeNewswire (Europe) | Initial AlloNK® (AB-101) clinical data demonstrated 71% ACR50 response in refractory rheumatoid arthritis (RA) patients with at least six months of follow-up in the company-sponsored Phase 2a basket... ► Artikel lesen | |
| 08.05. | Artiva Biotherapeutics, Inc.: Artiva Announces Positive Initial Clinical Data with AlloNK Across Multiple Autoimmune Diseases and FDA Alignment to Initiate Phase 3 Registrational Trial in Rheumatoid Arthritis in 2026 | 262 | GlobeNewswire (Europe) | Initial clinical data demonstrated 71% ACR50 response in refractory rheumatoid arthritis (RA) patients with at least six months of follow-up in the company-sponsored Phase 2a basket trial, with no... ► Artikel lesen | |
| 08.05. | Artiva Biotherapeutics, Inc. - 10-Q, Quarterly Report | - | SEC Filings | ||
| 08.05. | Artiva Biotherapeutics, Inc. - 8-K, Current Report | - | SEC Filings | ||
| 11.03. | Cantor Fitzgerald reiterates Artiva Biotherapeutics stock rating | 2 | Investing.com |
1 2 Weiter >>
32 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 77,05 | -1,34 % | BioNTech und BMS melden neue Daten für Lungenkrebskandidat Pumitamig | BioNTech SE und Bristol Myers Squibb haben auf dem ASCO-Jahreskongress in Chicago erste globale Phase-2-Daten für den bispezifischen Immunmodulator Pumitamig in Kombination mit Chemotherapie bei fortgeschrittenem... ► Artikel lesen | |
| EVOTEC | 4,932 | -3,58 % | EQS-News: Evotec SE: Evotec ernennt Rui Wang zum Head of Global In Silico & AI | EQS-News: Evotec SE
/ Schlagwort(e): Sonstiges
Evotec ernennt Rui Wang zum Head of Global In Silico & AI
02.06.2026 / 07:30 CET/CEST
Für den Inhalt der Mitteilung ist... ► Artikel lesen | |
| MODERNA | 41,190 | +0,12 % | Neue CAR-T-Erfolge bei Autoimmunerkrankungen: Warum Lösungen für Zelltherapien vor dem nächsten Wachstumsschub stehen könnten | ||
| VALNEVA | 2,331 | -0,64 % | Valneva im Höhenflug: Das Signal das die meisten Anleger noch nicht kennen - Meine Einschätzung für die neue Börsenwoche | ||
| AMGEN | 306,30 | +0,97 % | EQS-News: Amgen: Europäische Kommission erteilt Zulassung für Imdylltra von Amgen für die Behandlung von kleinzelligem Lungenkrebs im fortgeschrittenen Stadium | EQS-News: Amgen
/ Schlagwort(e): Sonstiges/Sonstiges
EUROPÄISCHE KOMMISSION ERTEILT ZULASSUNG FÜR IMDYLLTRA VON AMGEN FÜR DIE BEHANDLUNG VON KLEINZELLIGEM... ► Artikel lesen | |
| NOVAVAX | 8,080 | -1,70 % | NOVAVAX INC: Stabilität vor der nächsten Bewegung | ||
| BIOGEN | 171,56 | +1,19 % | Biogen's Salanersen Wins FDA Breakthrough Therapy Designation For Spinal Muscular Atrophy Treatment | WESTON (dpa-AFX) - Thursday, Biogen Inc. (BIIB) announced that the U.S. Food and Drug Administration has granted salanersen Breakthrough Therapy Designation for the treatment of spinal muscular... ► Artikel lesen | |
| PALATIN TECHNOLOGIES | 14,200 | +3,65 % | Palatin Technologies, Inc.: Palatin to Begin Trading on Nasdaq Stock Exchange | PRINCETON, N.J., May 18, 2026 /PRNewswire/ -- Palatin Technologies, Inc. (NYSE American: PTN), a biopharmaceutical company focused on the discovery and development... ► Artikel lesen | |
| VIVOSIM LABS | 1,210 | +0,83 % | VivoSim Labs, Inc.: VivoSim Appoints Arumugham (Ragoo) Raghunathan as Vice President of Global Sales | SAN DIEGO, March 03, 2026 (GLOBE NEWSWIRE) -- VivoSim Labs, Inc. (Nasdaq: VIVS) (the "Company" or "VivoSim"), a provider of next-generation New Approach Methodologies (NAMs) for preclinical safety... ► Artikel lesen | |
| SANGAMO THERAPEUTICS | 0,126 | -100,00 % | Sangamo Therapeutics, Inc.: Sangamo Therapeutics Reports Recent Business Highlights and First Quarter 2026 Financial Results | Rolling submission of Biologics License Agreement (BLA) to U.S. Food and Drug Administration (FDA) seeking ST-920 approval is in progress. Six sites activated in Phase 1/2 STAND study evaluating ST-503... ► Artikel lesen | |
| VIKING THERAPEUTICS | 24,650 | -0,20 % | XETR NEW INSTRUMENTS AVAILABLE ON XETRA - 04.06.2026 | Folgende Wertpapiere werden in den Handel an folgenden Handelsplätzen (MIC Code) im Quotation Board an der Frankfurter Wertpapierbörse (FWB) aufgenommen. Die Notierung an anderen Handelsplätzen im Quotation... ► Artikel lesen | |
| INTELLIA THERAPEUTICS | 11,240 | -4,26 % | Intellia Therapeutics, Inc. (NTLA) Buy Rating Reiterated Despite Share Offering Dilution | ||
| TEMPUS AI | 40,195 | -0,22 % | Tempus AI gründet Open-Source-Konsortium für digitale Pathologie | ||
| BIOCRYST PHARMACEUTICALS | 7,232 | -0,85 % | BioCryst Ireland, a Neopharmed Gentili company: Hereditary angioedema, a European initiative to shine a spotlight on a still under-recognized condition | Ahead of World HAE Day, BioCryst Ireland, a Neopharmed Gentili company, organised an international event in Milan bringing together leading experts from across Europe to raise awareness of... ► Artikel lesen | |
| BIOMARIN PHARMACEUTICAL | 49,500 | +0,49 % | BIOMARIN PHARMACEUTICAL INC - 8-K, Current Report |
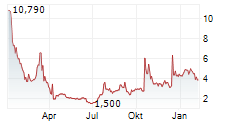
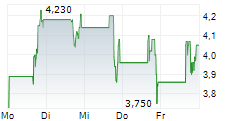
ARTIVA BIOTHERAPEUTICS INC gehört der Branche Biotechnologie an. Mit einer aktuellen Kursveränderung von -0,030 (-0,44 %) liegt der Kurs bei 6,820 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu ARTIVA BIOTHERAPEUTICS INC finden Sie auf Wallstreet Online.