| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
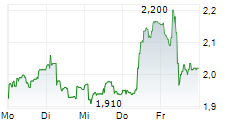
| 2,065 | 2,095 | 02.06. | |
| 2,040 | 2,075 | 02.06. |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 22.05. | CROSSJECT announces the successful completion of an equity financing and warrant issuance for a total immediate gross amount of €15 million | 211 | GlobeNewswire (Europe) | Provides immediate new financing to support the Company's next strategic milestones Strengthens shareholders' equity and improves the Company's financial structure Supports the continuation of ZEPIZURE®... ► Artikel lesen | |
| 25.03. | CROSSJECT appoints Lionel Seltz as Chief Financial Officer | 290 | GlobeNewswire (Europe) | DIJON, France - 25 mars 2026 (06.00 PM CET) - CROSSJECT (ISIN: FR0011716265; Euronext: ALCJ), the specialty pharmaceuticals company developing emergency medicines based on its proprietary ZENEO®... ► Artikel lesen | |
| 25.03. | CROSSJECT publishes its 2025 financial results and confirms the strengthening of its operational, industrial and financial trajectory | 534 | GlobeNewswire (Europe) | DIJON, France - 25 March 2026 (07:20 AM CET) - CROSSJECT (ISIN: FR0011716265; Euronext: ALCJ), a specialty pharmaceutical company developing products for emergency situations based on its proprietary... ► Artikel lesen | |
| CROSSJECT Aktie jetzt für 0€ handeln | |||||
| 02.02. | CROSSJECT announces initiation of coverage of its stock by Portzamparc (BNP Paribas Group) | 763 | GlobeNewswire (Europe) | CROSSJECT announces initiation of coverage of its stock by Portzamparc (BNP Paribas Group) DIJON, France - February 2, 2026 (7:15 a.m. CET) - CROSSJECT (ISIN: FR0011716265; Euronext: ALCJ), the specialty... ► Artikel lesen | |
| 14.11.25 | CROSSJECT completes €5 million financing with Vatel Capital | 668 | GlobeNewswire (Europe) | Press release CROSSJECT completes €5 million financing with Vatel Capital DIJON, France - November 14th, 2025 (8.30 PM CET) - CROSSJECT (ISIN: FR0011716265; Euronext: ALCJ), a specialty pharmaceuticals... ► Artikel lesen | |
| 24.09.25 | CROSSJECT presents its financial results and key highlights for the first half of 2025 | 371 | GlobeNewswire (Europe) | Press release CROSSJECT presents its financial results and key highlights for the first half of 2025 Cash position: €6.3 million, compared to €7.0 million as of December 31 2024, demonstrating the... ► Artikel lesen | |
| 22.09.25 | CROSSJECT Secures Additional BARDA Funding for the Progressive Developmental and FDA Authorization of the ZEPIZURE (ZENEO Midazolam) | 276 | GlobeNewswire (Europe) | Press release CROSSJECT Secures Additional BARDA Funding for the Progressive Developmental and FDA Authorization of the ZEPIZURE® (ZENEO® Midazolam) DIJON, France - September 22, 2025 (7.30 AM... ► Artikel lesen | |
| 11.06.25 | CROSSJECT advances on the development of ZEPIZURE Junior, its solution for epilepsy crises in children | 304 | GlobeNewswire (Europe) | Press release CROSSJECT advances on the development of ZEPIZURE® Junior, its solution for epilepsy crises in children Announces the effective calibration of ZENEO® for the use of ZEPIZURE® in the... ► Artikel lesen | |
| 03.06.25 | Maxim Group initiates the coverage of the CROSSJECT share with a BUY recommendation and a target price of €4.00 | 230 | GlobeNewswire (Europe) | Press release Maxim Group initiates the coverage of the CROSSJECT share with a BUY recommendation and a target price of €4.00 Dijon, France 03 June 2025 (07.30 CET) -- CROSSJECT (ISIN: FR0011716265... ► Artikel lesen |
9 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| FRESENIUS | 36,450 | +2,82 % | dpa-AFX-Überblick: UNTERNEHMEN vom 26.05.2026 - 15.15 Uhr | Schwerwiegende Bedenken: BP trennt sich von Verwaltungsratschef - Kursrutsch LONDON - Der britische Ölkonzern BP setzt seinen Verwaltungsratschef Albert Manifold wegen Vorwürfen vor die Tür. Als... ► Artikel lesen | |
| SIEMENS HEALTHINEERS | 34,040 | -0,64 % | EQS-CMS: Siemens Healthineers AG: Veröffentlichung einer Kapitalmarktinformation | EQS Zulassungsfolgepflichtmitteilung: Siemens Healthineers AG
/ Bekanntmachung nach Art. 5 Abs. 1 lit. a) der Verordnung (EU) Nr. 596/2014, zuletzt geändert durch die Verordnung (EU)... ► Artikel lesen | |
| DRAEGERWERK | 89,20 | -0,56 % | JEFFERIES stuft Draegerwerk auf 'Hold' | NEW YORK (dpa-AFX Analyser) - Das Analysehaus Jefferies hat das Kursziel für Drägerwerk von 73 auf 90 Euro angehoben, aber die Einstufung auf "Hold" belassen. Die Margenentwicklung des Spezialisten... ► Artikel lesen | |
| GERRESHEIMER | 26,120 | -3,55 % | EQS-PVR: Gerresheimer AG: Veröffentlichung gemäß § 40 Abs. 1 WpHG mit dem Ziel der europaweiten Verbreitung | EQS Stimmrechtsmitteilung: Gerresheimer AG
Gerresheimer AG: Veröffentlichung gemäß § 40 Abs. 1 WpHG mit dem Ziel der europaweiten Verbreitung
27.05.2026 / 18:42 CET/CEST
Veröffentlichung... ► Artikel lesen | |
| INTUITIVE SURGICAL | 344,60 | -2,78 % | Intuitive Surgical, Inc.: Intuitive Announces New Chief Commercial and Marketing Officer Taylor Patton | SUNNYVALE, Calif., May 28, 2026 (GLOBE NEWSWIRE) -- Intuitive (NASDAQ: ISRG), a global technology leader in minimally invasive care and the pioneer of robotic-assisted surgery, today announced the... ► Artikel lesen | |
| THERMO FISHER | 416,40 | -1,95 % | Laufende Fusionskontrollverfahren: Astorg Group S.à.r.l., Luxemburg; mittelbarer Erwerb alleiniger Kontrolle über den Mikrobiologie-Geschäftsbereich von Thermo Fisher Scientific Inc., Waltham/MA/USA (u.a. Oxoid Deutschland GmbH) | Datum der Anmeldung:22.05.2026Aktenzeichen:B3-68/26Unternehmen:Astorg Group S.à.r.l., Luxemburg; mittelbarer Erwerb alleiniger Kontrolle über den Mikrobiologie-Geschäftsbereich von Thermo Fisher Scientific... ► Artikel lesen | |
| HIMS & HERS HEALTH | 23,760 | -0,42 % | Telemedizin-Aktie legt zu: 1,15-Milliarden-Deal: Hims & Hers expandiert radikal | © Foto: picture alliance / ZUMAPRESS.com | Avishek DasHims & Hers kauft sich mit Eucalyptus in neue Märkte ein - der Milliarden-Deal schürt Fantasie für den nächsten Wachstumsschub.Hims & Hers Health... ► Artikel lesen | |
| SERNOVA BIOTHERAPEUTICS | 0,099 | +4,01 % | Sernova Biotherapeutics: Sernova Biotherapeutics Announces Closing of Recently Approved Financings | Toronto, Ontario and Boston, Massachusetts--(Newsfile Corp. - April 23, 2026) - Sernova Biotherapeutics (TSX: SVA) (OTC: SEOVF) (FSE: PSHO) (XETRA: PSHO) ("Sernova" or the "Company"), a leading... ► Artikel lesen | |
| FAMICORD | 3,910 | -1,26 % | EQS-News: FamiCord AG verzeichnet erwartungsgemäß schwächeren Start in das Geschäftsjahr 2026 bei anhaltendem Gegenwind aus dem Markt und robuster Abonnementbasis | EQS-News: FamiCord AG
/ Schlagwort(e): Quartalsergebnis
FamiCord AG verzeichnet erwartungsgemäß schwächeren Start in das Geschäftsjahr 2026 bei anhaltendem Gegenwind aus dem Markt... ► Artikel lesen | |
| GERATHERM MEDICAL | 2,710 | -3,21 % | Geratherm Medical wechselt vom Börsensegment Scale in den Open Market | Die Geratherm Medical AG beendet zum 27. April 2026 die Einbeziehung ihrer Aktien in das Börsensegment Scale der Frankfurter Wertpapierbörse und wechselt ins Basic Board am Open Market. Die Börsennotierung... ► Artikel lesen | |
| ALIGN TECHNOLOGY | 144,85 | -0,75 % | Align Technology, Inc.: Align Technology Awards Funding to Universities Worldwide for Advancing Orthodontic and Dental Research | $300,000 in Research Grants Awarded as Part of its Ongoing Annual Research Awards Program
Align Technology, Inc. ("Align") (Nasdaq: ALGN), a leading global medical device company that designs,... ► Artikel lesen | |
| OTTOBOCK | 53,10 | +1,14 % | Ottobock bringt 3D-gedruckten Silikon-Liner für Beinprothesen auf den Markt | ||
| NUGEN MEDICAL DEVICES | 0,012 | -21,52 % | NuGen Medical Devices Inc.: NuGen Medical Devices Files Patent for Dual Spring Technology Enhancing Needle-Free Injection Performance | Toronto, Ontario--(Newsfile Corp. - May 28, 2026) - NuGen Medical Devices Inc. (TSXV: NGMD) ("NuGen" or the "Company"), a leader in needle free drug delivery, today announced the filing of a new... ► Artikel lesen | |
| PLUS THERAPEUTICS | 4,805 | -1,84 % | Plus Therapeutics Inc.: Plus' CNSide Diagnostic Platform Demonstrates Substantial Healthcare Cost Reduction in Patients with Leptomeningeal Metastases | ||
| CARDINAL HEALTH | 166,95 | -0,51 % | Neun Dividendenaufstockungen im Fokus: Was die jüngsten Erhöhungen von Cardinal Health, Constellation Energy & Co. für Einkommensinvestoren bedeuten | Mehrere US-Unternehmen haben Anfang Mai ihre Dividenden angehoben - darunter mit Cardinal Health und Constellation Energy zwei prominente Vertreter aus Gesundheitswesen und Versorgersektor. Die von... ► Artikel lesen |
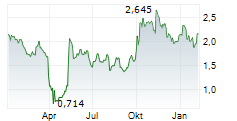
Das Unternehmen CROSSJECT SA kann der Branche Gesundheitswesen zugeordnet werden. Mit einer aktuellen Kursveränderung von -0,125 (-5,66 %) liegt der Kurs bei 2,085 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu CROSSJECT SA finden Sie auf Wallstreet Online.