- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 27.05. | Ernexa Therapeutics treibt IND-Antrag für Zelltherapie-Kandidaten ERNA-101 für Q3 voran | 1 | Investing.com Deutsch | ||
| 27.05. | Ernexa Therapeutics advances ERNA-101 toward Q3 IND filing | 2 | Investing.com | ||
| 27.05. | Ernexa Therapeutics Inc.: Ernexa Therapeutics Successfully Completes Key Manufacturing and IND Preparation Milestones for ERNA-101 Toward First-in-Human Clinical Development | 113 | GlobeNewswire (Europe) | CAMBRIDGE, Mass., May 27, 2026 (GLOBE NEWSWIRE) -- Ernexa Therapeutics (Nasdaq: ERNA), an industry innovator developing novel cell therapies for the treatment of advanced cancer and autoimmune disease... ► Artikel lesen | |
| 20.05. | Ernexa Therapeutics Inc.: Ernexa Therapeutics Releases Virtual Investor KOL Connect Segment Featuring Dr. Elena Ratner Highlighting Ovarian Cancer Landscape and ERNA-101 Opportunity | 1 | GlobeNewswire (USA) | ||
| 11.05. | Ernexa Therapeutics Inc. - 10-Q, Quarterly Report | 1 | SEC Filings | ||
| 11.05. | Ernexa Therapeutics Inc.: Ernexa Therapeutics Releases Virtual Investor KOL Connect Segment Highlighting Ovarian Cancer Landscape and Lead Candidate ERNA-101 | 2 | GlobeNewswire (USA) | ||
| 06.05. | Ernexa-Aktie schießt nach Studienergebnissen zu Eierstockkrebs um 40 % in die Höhe | 10 | Investing.com Deutsch | ||
| ERNEXA THERAPEUTICS Aktie jetzt für 0€ handeln | |||||
| 06.05. | Ernexa Therapeutics Inc.: Ernexa Therapeutics Unveils Breakthrough Preclinical Results: ERNA-101 Achieves 100% Survival and Complete Tumor Elimination in Ovarian Cancer Models | 290 | GlobeNewswire (Europe) | CAMBRIDGE, Mass., May 06, 2026 (GLOBE NEWSWIRE) -- Ernexa Therapeutics (Nasdaq: ERNA), an industry innovator developing novel cell therapies for the treatment of advanced cancer and autoimmune disease... ► Artikel lesen | |
| 04.05. | Ernexa Therapeutics Inc. - 8-K, Current Report | - | SEC Filings | ||
| 30.04. | Ernexa Therapeutics announces 1-for-25 reverse stock split; shares down | 2 | Seeking Alpha | ||
| 30.04. | Ernexa Therapeutics Inc.: Ernexa Therapeutics Announces 1-for-25 Reverse Stock Split | 186 | GlobeNewswire (Europe) | CAMBRIDGE, Mass., April 30, 2026 (GLOBE NEWSWIRE) -- Ernexa Therapeutics (Nasdaq: ERNA), an industry innovator developing novel cell therapies for the treatment of advanced cancer and autoimmune disease... ► Artikel lesen | |
| 31.03. | Ernexa plant klinische Studie für Zelltherapie ERNA-101 bei Eierstockkrebs | 1 | Investing.com Deutsch | ||
| 31.03. | Ernexa plans ovarian cancer trial for cell therapy ERNA-101 | 1 | Investing.com | ||
| 31.03. | Ernexa Therapeutics Inc.: Ernexa Therapeutics Highlights Key Catalysts as Company Advances Toward First-in-Human Clinical Trials in 2026 | 170 | GlobeNewswire (Europe) | Preclinical data demonstrate deep and durable tumor regressions with lead product candidate ERNA-101 in combination with PD-1 Blockade in ovarian cancer IND submission for ERNA-101 expected in Q3... ► Artikel lesen | |
| 20.03. | NASDAQ warnt Ernexa Therapeutics vor Delisting wegen niedrigem Aktienkurs | 4 | Investing.com Deutsch | ||
| 20.03. | Ernexa Therapeutics Inc. - 8-K, Current Report | 2 | SEC Filings | ||
| 13.03. | Ernexa Therapeutics Inc. - 10-K, Annual Report | 2 | SEC Filings | ||
| 26.02. | Ernexa Therapeutics Inc. - 8-K, Current Report | 3 | SEC Filings | ||
| 20.02. | Ernexa Therapeutics Inc.: Ernexa Therapeutics to Participate in Virtual Investor Closing Bell Series | 1 | GlobeNewswire (USA) | ||
| 11.02. | Ernexa Therapeutics Inc. - 8-K, Current Report | 1 | SEC Filings |
1 2 Weiter >>
34 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| EVOTEC | 5,025 | -1,37 % | EQS-NVR: Evotec SE: Veröffentlichung der Gesamtzahl der Stimmrechte nach § 41 WpHG mit dem Ziel der europaweiten Verbreitung | EQS Gesamtstimmrechtsmitteilung: Evotec SE
/ Veröffentlichung der Gesamtzahl der Stimmrechte
Evotec SE: Veröffentlichung der Gesamtzahl der Stimmrechte nach § 41 WpHG mit dem... ► Artikel lesen | |
| MODERNA | 41,480 | +5,75 % | Neue CAR-T-Erfolge bei Autoimmunerkrankungen: Warum Lösungen für Zelltherapien vor dem nächsten Wachstumsschub stehen könnten | ||
| VALNEVA | 2,381 | -2,18 % | Original-Research: Valneva SE (von First Berlin Equity Research GmbH): Buy | Original-Research: Valneva SE - von First Berlin Equity Research GmbH
28.05.2026 / 17:08 CET/CEST
Veröffentlichung einer Research, übermittelt durch EQS News - ein Service der EQS Group.
Für den... ► Artikel lesen | |
| PALATIN TECHNOLOGIES | 13,500 | -3,98 % | PALATIN TECHNOLOGIES INC - 8-A12B, Registration of securities | ||
| VIVOSIM LABS | 1,250 | -3,10 % | VivoSim Labs, Inc.: VivoSim Appoints Arumugham (Ragoo) Raghunathan as Vice President of Global Sales | SAN DIEGO, March 03, 2026 (GLOBE NEWSWIRE) -- VivoSim Labs, Inc. (Nasdaq: VIVS) (the "Company" or "VivoSim"), a provider of next-generation New Approach Methodologies (NAMs) for preclinical safety... ► Artikel lesen | |
| SANGAMO THERAPEUTICS | 0,126 | -100,00 % | Sangamo Therapeutics, Inc.: Sangamo Therapeutics Reports Recent Business Highlights and First Quarter 2026 Financial Results | Rolling submission of Biologics License Agreement (BLA) to U.S. Food and Drug Administration (FDA) seeking ST-920 approval is in progress. Six sites activated in Phase 1/2 STAND study evaluating ST-503... ► Artikel lesen | |
| INTELLIA THERAPEUTICS | 11,140 | -1,42 % | Intellia to present Phase 3 gene editing trial data at congress | ||
| ARBUTUS BIOPHARMA | 3,630 | -1,09 % | Arbutus Biopharma Corp - 8-K, Current Report | ||
| XOMA ROYALTY | 35,600 | -1,11 % | Weekly Buzz: FDA Okays AstraZeneca's Saphnelo Pen, EC Clears Crinetics Pharmaceuticals' Acromegaly Drug, Soligenix Slumps, Ligand Snaps Up XOMA Royalty | BASEL (dpa-AFX) - This week's biotech landscape witnessed regulatory approvals in the US and Europe, acquisitions, and a few trial halts, alongside positive data readouts spanning multiple therapeutic... ► Artikel lesen | |
| CELLECTAR BIOSCIENCES | 2,630 | -4,36 % | Cellectar Biosciences, Inc.: Cellectar Biosciences to Highlight Compelling New Efficacy Data from Phase 2b CLOVER WaM Study of Iopofosine I 131 in r/r Waldenström Macroglobulinemia at the American Society of Clinical Oncology Annual Meeting | Approximately 80% Major Response Rate and Duration of Response Exceeding 16 Months in Subset Analysis of Patients Treated with Iopofosine I 131 Immediately Post-BTKi Therapy Plans to Initiate Phase... ► Artikel lesen | |
| AIM IMMUNOTECH | 0,073 | -100,00 % | AIM ImmunoTech Inc.: AIM ImmunoTech Completes Phase 2 DURIPANC Enrollment Milestone Ahead of Schedule, Advancing Ampligen Toward Critical Pancreatic Cancer Clinical Milestones | OCALA, Fla., June 01, 2026 (GLOBE NEWSWIRE) -- AIM ImmunoTech Inc. (NYSE American: AIM) ("AIM" or the "Company") today announced a significant milestone in its pancreatic cancer development program... ► Artikel lesen | |
| NOVOCURE | 13,195 | -4,90 % | Novocure Reports First Quarter 2026 Financial Results | Quarterly net revenues of $174 million, up 12% year-over-year, driven by global growth in Optune Gio
Optune Pax approved by the U.S. FDA and launched for the treatment of locally advanced pancreatic... ► Artikel lesen | |
| PHARMING | 1,064 | -0,51 % | Pharming Group N.V.: Pharming Group to participate in Fireside Chat at 2026 Jefferies Global Healthcare Conference in New York | Leiden, the Netherlands, June 2, 2026: Pharming Group ("Pharming") (Euronext Amsterdam: PHARM/Nasdaq: PHAR) today announced its participation in the 2026 Jefferies Global Healthcare Conference in New... ► Artikel lesen | |
| CYTOKINETICS | 60,50 | -0,79 % | Cytokinetics, Incorporated: Cytokinetics bringt MYQORZO (aficamten) in der Europäischen Union auf den Markt; der erste kommerzielle Vertrieb erfolgt in Deutschland | Auf den erfolgreichen Start in den USA folgt nun die Markteinführung in Europa
SOUTH SAN FRANCISCO, Kalifornien, und ZUG, Schweiz, June 01, 2026 (GLOBE NEWSWIRE) -- Cytokinetics, Incorporated (Nasdaq:... ► Artikel lesen | |
| TRAWS PHARMA | 1,460 | +2,82 % | Traws Pharma, Inc.: Traws Pharma Announces Antiviral Program Targeting Ongoing Hantavirus and Ebola Virus Disease Outbreaks | Broad-spectrum antiviral agent with potential utility against Hantavirus, Ebola and Lassa Utilizing a novel dual-strike mechanism, the proprietary combination agent has the potential to deliver a... ► Artikel lesen |
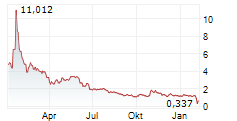
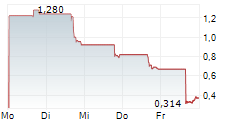
Das Unternehmen ERNEXA THERAPEUTICS INC kann der Branche Biotechnologie zugeordnet werden. Mit einer aktuellen Kursveränderung von +0,150 (+1,88 %) liegt der Kurs bei 8,150 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu ERNEXA THERAPEUTICS INC finden Sie auf Wallstreet Online.