- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| Do | Fortress Biotech, Inc. - 10-Q, Quarterly Report | 1 | SEC Filings | ||
| Do | Fortress Biotech GAAP EPS of $2.82, revenue of $16.04M misses by $0.3M | 3 | Seeking Alpha | ||
| Do | Fortress Biotech, Inc. - 8-K, Current Report | 1 | SEC Filings | ||
| Do | Fortress Biotech, Inc.: Fortress Biotech Reports First Quarter 2026 Financial Results and Recent Corporate Highlights | 443 | GlobeNewswire (Europe) | ZYCUBO approved by FDA to treat Menkes disease in the United States; Fortress subsidiary Cyprium Therapeutics closed the sale of its Rare Pediatric Disease Priority Review Voucher (PRV) for $205 million... ► Artikel lesen | |
| 31.03. | Fortress Biotech GAAP EPS of -$0.07 misses by $0.39, revenue of $63.26M misses by $10.08M | 6 | Seeking Alpha | ||
| 31.03. | Fortress Biotech, Inc.: Fortress Biotech Reports 2025 Financial Results and Recent Corporate Highlights | 723 | GlobeNewswire (Europe) | ZYCUBOapproved by FDA to treat Menkes disease in the United States; Fortress subsidiary Cyprium Therapeutics sold its Rare Pediatric Disease Priority Review Voucher (PRV) for gross proceeds of $205... ► Artikel lesen | |
| FORTRESS BIOTECH Aktie jetzt für 0€ handeln | |||||
| 31.03. | Fortress Biotech, Inc. - 10-K, Annual Report | - | SEC Filings | ||
| 30.03. | Fortress Biotech, Inc.: Fortress Biotech's Subsidiary Cyprium Therapeutics Closes Sale of Rare Pediatric Disease Priority Review Voucher for $205 Million | 350 | GlobeNewswire (Europe) | MIAMI, March 30, 2026 (GLOBE NEWSWIRE) -- Fortress Biotech, Inc. (Nasdaq: FBIO) ("Fortress") and its majority-owned subsidiary, Cyprium Therapeutics, Inc. ("Cyprium"), today announced the closing... ► Artikel lesen | |
| 23.02. | Fortress Biotech, Inc.: Fortress Biotech's Subsidiary Cyprium Therapeutics Enters into Agreement to Sell Rare Pediatric Disease Priority Review Voucher for $205 Million | 452 | GlobeNewswire (Europe) | MIAMI, Feb. 23, 2026 (GLOBE NEWSWIRE) -- Fortress Biotech, Inc. (Nasdaq: FBIO) ("Fortress") and its majority-owned subsidiary, Cyprium Therapeutics, Inc. ("Cyprium"), today announced that Cyprium... ► Artikel lesen | |
| 23.02. | Fortress Biotech, Inc. - 8-K, Current Report | 5 | SEC Filings | ||
| 16.01. | Weekly Buzz: Lyra Therapeutics Slashes Jobs; FDA Okays Fortress Biotech's ZYCUBO; Boston Scientific To Acquire Penumbra; BriaCell Therapeutics On Watch | 814 | AFX News | NEW BRUNSWICK (dpa-AFX) - This week, the biotech sector saw a steady stream of high-impact news, including a major workforce reduction and a fresh round of regulatory clearances spanning diabetes... ► Artikel lesen | |
| 14.01. | Fortress Biotech receives FDA approval for Zycubo to treat Menkes disease | 13 | Pharmaceutical Technology | ||
| 13.01. | Fortress Bio gains on FDA approval of copper replacement therapy | 6 | Seeking Alpha | ||
| 13.01. | Fortress Biotech, Inc. - 8-K, Current Report | 2 | SEC Filings | ||
| 13.01. | Fortress Biotech, Inc.: Fortress Biotech and Cyprium Therapeutics Announce U.S. FDA Approval of ZYCUBO (copper histidinate), the First and Only Approved Treatment for Menkes Disease in the United States | 872 | GlobeNewswire (Europe) | Rare Pediatric Disease Priority Review Voucher (PRV) granted by FDA at approval to be transferred from Sentynl Therapeutics to Cyprium Cyprium eligible to receive tiered royalties and up to $129 million... ► Artikel lesen | |
| 15.12.25 | FDA sets new PDUFA date for Fortress Biotech's Menkes disease therapy | 31 | Investing.com | ||
| 15.12.25 | Fortress Biotech, Inc. - 8-K, Current Report | 3 | SEC Filings | ||
| 14.11.25 | Fortress Biotech, Inc.: Fortress Biotech Reports Third Quarter 2025 Financial Results and Recent Corporate Highlights | 760 | GlobeNewswire (Europe) | Total net revenue increased 20.5% to $17.6 million for third quarter of 2025 compared to the third quarter of 2024 Fortress subsidiary Checkpoint Therapeutics acquired by Sun Pharma; Fortress received... ► Artikel lesen | |
| 21.10.25 | Fortress Biotech, Inc.: Fortress Biotech and Subsidiary Urica Therapeutics Announce First Patients Dosed in Crystalys Therapeutics' Global Phase 3 Trials of Dotinurad for the Treatment of Gout | 347 | GlobeNewswire (Europe) | MIAMI, Oct. 21, 2025 (GLOBE NEWSWIRE) -- Urica Therapeutics, Inc. ("Urica" or the "Company"), a Fortress Biotech, Inc. (Nasdaq: FBIO) ("Fortress") subsidiary, today announced that Crystalys Therapeutics... ► Artikel lesen | |
| 01.10.25 | Fortress Biotech, Inc.: Fortress Biotech and Subsidiary Urica Therapeutics Announce Crystalys Therapeutics' $205 Million Series A Financing | 207 | GlobeNewswire (Europe) | Dotinurad is a next-generation URAT1 inhibitor in two Phase 3 clinical trials with potential for best-in-class safety and efficacy Urica sold dotinurad to Crystalys Therapeutics in 2024 in exchange... ► Artikel lesen |
1 2 Weiter >>
23 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 77,75 | +2,71 % | Warum Biotech-Aktien jetzt im Umbruch sind und Zelltherapie nicht nur bei BioNTech, Morphosys und Co. ein Thema ist. | ||
| EVOTEC | 4,832 | +1,94 % | Evotec kann nicht alle Wandelanleihen platzieren | Das Hamburger Biotech-Unternehmen Evotec hat eine Wandelanleihe mit einem Gesamtnennbetrag von 116,1 Millionen Euro platziert - nach einem ursprünglich angekündigten Volumen von bis zu 125 Millionen... ► Artikel lesen | |
| BB BIOTECH | 47,950 | +0,95 % | Aktien KW 17 Märkte im Würgegriff. Planloser Trump , selbstbewusterer Iran. Lösungen müssen her. News. Rheinmetall. ITM Power. Porsche. The Platform Group. K+S. Bugatti. Circus. Surteco. NanoRepro. BB Biotech. Mensch und Maschine. ATOSS. MHP Hotel | Aktien - letzte Woche warteten die Märkte auf eine Lösung des Iran-Kriegs. Aber die Iraner sehen sich im Vorteil und Trump verlängert "auf unbestimmte Zeit" den Waffenstillstand. Aber die Strasse von... ► Artikel lesen | |
| MEDIGENE | 0,026 | -10,27 % | PTA-Adhoc: Medigene AG: Widerruf der Zulassung der Aktien der Medigene AG zum Handel im regulierten Markt | DJ PTA-Adhoc: Medigene AG: Widerruf der Zulassung der Aktien der Medigene AG zum Handel im regulierten Markt
Veröffentlichung von Insiderinformationen gemäß Artikel 17 MAR Schlagwort(e): Sonstiges... ► Artikel lesen | |
| QIAGEN | 29,745 | +0,10 % | QIAGEN N.V.: QIAGEN to advance AI-driven drug discovery with graph-based AI and curated bioinformatics knowledge with NVIDIA | Collaboration integrates NVIDIA accelerated computing and the NVIDIA BioNeMo platform with QIAGEN Digital Insights' curated biomedical knowledge bases Goal to advance how disease mechanisms... ► Artikel lesen | |
| MODERNA | 41,780 | +6,07 % | Hedge Fund and Insider Trading News: Bill Ackman, Ray Dalio, Warren Buffett, Viking Global, Darsana Capital Partners, Caxton Associates, Balyasny Asset Management, D1 Capital Partners, Republic Services Inc (RSG), Moderna Inc (MRNA), and More | ||
| PAION | 0,072 | -25,52 % | XFRA MISTRADE ANTRAG IN ISIN DE000A3E5EG5 WURDE STATTGEGEBEN | Dem Mistrade-Antrag wurde stattgegeben. Folgende Geschaefte in der ISIN DE000A3E5EG5, Wertpapier-Name: PAION AG INH O.N., wurden aufgehoben:ISIN Datum Zeit Volumen PreisDE000A3E5EG5 02.02.2026 08:13:40 300 0... ► Artikel lesen | |
| VALNEVA | 2,480 | +1,56 % | Valneva senkt Prognose und baut Stellen ab | Beim Impfstoffspezialisten Valneva SE häufen sich im ersten Quartal 2026 die Belastungen: Der Nettoverlust weitete sich auf 32,1 Millionen Euro nach 9,2 Millionen Euro im Vorjahr aus, das bereinigte... ► Artikel lesen | |
| AMGEN | 283,70 | -0,46 % | Dividendenbekanntmachungen (15.05.2026) | Unternehmen ISIN-Code Dividende (Währung) Dividende (EUR) A10 NETWORKS INC US0021211018 0,06 USD 0,0514 EUR AF GRUPPEN ASA NO0003078107 6,5 NOK 0,6029 EUR AGCO CORPORATION US0010841023 0... ► Artikel lesen | |
| EPIGENOMICS | 0,930 | -0,53 % | PTA-HV: Epigenomics AG: Einladung zur ordentlichen Hauptversammlung | DJ PTA-HV: Epigenomics AG: Einladung zur ordentlichen Hauptversammlung
Hauptversammlung gemäß -- 121 Abs. 4a AktG
Epigenomics AG: Einladung zur ordentlichen Hauptversammlung
Heidelberg... ► Artikel lesen | |
| NOVAVAX | 8,000 | +4,17 % | Novavax, Inc.: Novavax Reports First Quarter 2026 Financial Results and Operational Highlights | Total revenue of $140 million in the first quarter of 2026
Pfizer agreement announced in January for non-exclusive license to utilize Matrix-M® in two infectious... ► Artikel lesen | |
| STRYKER | 273,80 | +0,51 % | Stryker Buys Amplitude Vascular Systems For Upto $835 Mln To Expand Vascular Treatment Business | WASHINGTON (dpa-AFX) - Tuesday, BioStar Capital announced that Amplitude Vascular Systems, Inc., a medical technology company, has been acquired by Stryker Corporation (SYK) at a purchase price... ► Artikel lesen | |
| BIOGEN | 164,00 | -0,12 % | Biogen Inc.: Biogen Completes Acquisition of Apellis Pharmaceuticals | CAMBRIDGE, Mass., May 14, 2026 (GLOBE NEWSWIRE) -- Biogen Inc. (Nasdaq: BIIB) today announced the successful completion of the acquisition of Apellis Pharmaceuticals, Inc. (Nasdaq: APLS). Apellis... ► Artikel lesen | |
| BIOFRONTERA | 2,390 | -2,85 % | Biofrontera stock rating cut to Speculative Buy by Benchmark | ||
| HEIDELBERG PHARMA | 2,720 | -1,09 % | EQS-HV: Heidelberg Pharma AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in Ladenburg mit dem Ziel der europaweiten Verbreitung gemäß §121 AktG | EQS-News: Heidelberg Pharma AG
/ Bekanntmachung der Einberufung zur Hauptversammlung
Heidelberg Pharma AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in... ► Artikel lesen |
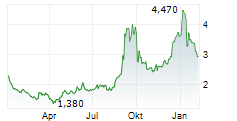
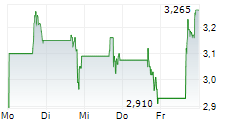
FORTRESS BIOTECH INC gehört der Branche Biotechnologie an. Der aktuelle Kurs liegt bei 2,665 Euro und damit +5,34 % im Plus.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu FORTRESS BIOTECH INC finden Sie auf Wallstreet Online.