- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 25.02. | Modus Therapeutics Holding AB: Modus Therapeutics publishes year-end report for 2025 | 1.124 | GlobeNewswire (Europe) | STOCKHOLM, SWEDEN - 25 February 2026: Modus Therapeutics Holding AB ("Modus Therapeutics") hereby publishes year-end report for 2025. The report is available on the company's website (www.modustx.com)... ► Artikel lesen | |
| 10.12.25 | Karolinska Development AB (publ): Karolinska Development's portfolio company Modus Therapeutics initiates dosing in phase 2a study in patients with chronic kidney disease with anemia | 271 | GlobeNewswire (Europe) | STOCKHOLM, SWEDEN December 10, 2025. Karolinska Development AB (Nasdaq Stockholm: KDEV) today announces that its portfolio company, Modus Therapeutics, has dosed the first patient in the phase 2a... ► Artikel lesen | |
| 26.11.25 | Modus Therapeutics Holding AB: Modus Therapeutics publishes Interim report for the third quarter 2025 | 488 | GlobeNewswire (Europe) | STOCKHOLM, SWEDEN - 26 November 2025: Modus Therapeutics Holding AB ("Modus Therapeutics") hereby publishes an interim report for the third quarter of 2025. The report is available on the company's... ► Artikel lesen | |
| 04.11.25 | Karolinska Development AB (publ): Karolinska Development's portfolio company Modus Therapeutics receives approval for second part of phase 2 study in chronic kidney disease with anemia | 276 | GlobeNewswire (Europe) | STOCKHOLM, SWEDEN November 4, 2025. Karolinska Development AB (Nasdaq Stockholm: KDEV) today announces that its portfolio company, Modus Therapeutics, has received regulatory approval to initiate... ► Artikel lesen | |
| MODUS THERAPEUTICS Aktie jetzt für 0€ handeln | |||||
| 03.11.25 | Modus Therapeutics Holding AB: Modus Therapeutics receives regulatory approval for Part 2 of the Phase IIa study in CKD-with anemia | 292 | GlobeNewswire (Europe) | Stockholm, Sweden - November 3, 2025- Modus Therapeutics Holding AB (publ) ("Modus" or the "Company") announces that the competent authorities in Italy have approved the Company's dose selection for... ► Artikel lesen | |
| 28.08.25 | Karolinska Development AB (publ): Karolinska Development's portfolio company Modus Therapeutics raises SEK 28.3 million in oversubscribed unit issue | 203 | GlobeNewswire (Europe) | STOCKHOLM, SWEDEN August 28, 2025. Karolinska Development AB (Nasdaq Stockholm: KDEV) today announces that its portfolio company, Modus Therapeutics, has raised SEK 28.3 million in a unit issue with... ► Artikel lesen | |
| 07.08.25 | Modus Therapeutics Holding AB: Modus Therapeutics publishes Interim report for the second quarter 2025 | 449 | GlobeNewswire (Europe) | STOCKHOLM, SWEDEN - 7 August 2025: Modus Therapeutics Holding AB ("Modus Therapeutics") hereby publishes an interim report for the second quarter of 2025. The report is available on the company's website... ► Artikel lesen | |
| 06.08.25 | XFRA CAPITAL ADJUSTMENT INFORMATION - 06.08.2025 | 559 | Xetra Newsboard | Das Instrument 9YG NO0010955917 DEEP VAL.DRILL.AS NK -,10 EQUITY wird cum Kapitalmassnahme gehandelt am 06.08.2025 und ex Kapitalmassnahme am 07.08.2025 The instrument 9YG NO0010955917 DEEP VAL.DRILL.AS... ► Artikel lesen | |
| 01.08.25 | Modus Therapeutics Holding AB: Modus Therapeutics on track to initiate Part 2 of Phase IIa sevuparin study in CKD-related anemia | 416 | GlobeNewswire (Europe) | Stockholm, Sweden - 1 August 2025 - Modus Therapeutics Holding AB (publ) ("Modus") today announces that dose levels for Part 2 of its ongoing Phase IIa clinical study of sevuparin in patients with chronic... ► Artikel lesen | |
| 29.07.25 | Modus Therapeutics Holding AB: Extraordinary General Meeting in Modus Therapeutics Holding AB (publ) | 272 | GlobeNewswire (Europe) | Modus Therapeutics' Extraordinary General Meeting on 29 July 2025 resolved in accordance with the following:
To amend the company's articles of association in order to increase the limits of the company's... ► Artikel lesen | |
| 14.07.25 | Karolinska Development AB (publ): Karolinska Development's portfolio company Modus Therapeutics completes enrollment in part 1 of its phase 2a study with sevuparin | 430 | GlobeNewswire (Europe) | STOCKHOLM, SWEDEN July 14, 2025. Karolinska Development AB (Nasdaq Stockholm: KDEV) today announces that its portfolio company Modus Therapeutics has completed patient enrollment on schedule to part... ► Artikel lesen | |
| 08.07.25 | Modus Therapeutics Holding AB: Modus Therapeutics completes enrollment in Part 1 of the Phase IIa sevuparin study in chronic kidney disease with anemia | 324 | GlobeNewswire (Europe) | Stockholm, Sweden - 8 July 2025 - Modus Therapeutics Holding AB (publ) ("Modus") today announces that patient enrollment in Part 1 of its ongoing Phase IIa study evaluating sevuparin for the treatment... ► Artikel lesen | |
| 27.06.25 | Karolinska Development AB (publ): Karolinska Development's portfolio company Modus Therapeutics carries out a fully secured rights issue of SEK 28.3 million | 129 | GlobeNewswire (Europe) | STOCKHOLM, SWEDEN June 27, 2025. Karolinska Development AB (Nasdaq Stockholm: KDEV) today announces that its portfolio company, Modus Therapeutics carries out a fully secured rights issue of units... ► Artikel lesen |
13 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| NANOREPRO | 1,650 | 0,00 % | NanoRepro: Mit Elan gestartet | "Wir sind auf einem guten Weg", sagt Lisa Jüngst, CEO von NanoRepro, am Ende ihrer Präsentation auf der Frühjahrskonferenz in Frankfurt. Die Aufgabenstellung könnte dabei ambitionierter kaum sein. Immerhin... ► Artikel lesen | |
| MARINOMED BIOTECH | 9,000 | -2,17 % | EQS-News: Marinomed Biotech AG gibt strategische Partnerschaft und weltweite Lizenzierung der Marinosolv-Technologie im Bereich Inhalationstherapie bekannt | EQS-News: Marinomed Biotech AG
/ Schlagwort(e): Kooperation
Marinomed Biotech AG gibt strategische Partnerschaft und weltweite Lizenzierung der Marinosolv-Technologie im Bereich... ► Artikel lesen | |
| REPLENISH NUTRIENTS | 0,071 | 0,00 % | Replenish Nutrients Holding GAAP EPS of -$0.03, revenue of $2.96M | ||
| SANA BIOTECHNOLOGY | 3,230 | +2,54 % | Sana Biotechnology, Inc: Sana Biotechnology Announces Sale of Approximately $69 Million of Shares Through its At-the-Market (ATM) Facility | SEATTLE, May 15, 2026 (GLOBE NEWSWIRE) -- Sana Biotechnology, Inc. (NASDAQ: SANA), a company focused on changing the possible for patients through engineered cells, today announced that it has sold... ► Artikel lesen | |
| NOVABRIDGE BIOSCIENCES | 1,460 | -5,19 % | NovaBridge Biosciences: NovaBridge Reports Full Year 2025 Financial Results and Provides Business Update | Successful transformation to global biotech platform with two mid-stage potential best-in-class/first-in-class programs, givastomig and VIS-101, with compelling proof-of-concept dataGivastomig, a... ► Artikel lesen | |
| GENEURO | 0,000 | -100,00 % | XFRA DELETION OF INSTRUMENTS FROM BOERSE FRANKFURT - 18.05.2026 | The following instruments on Boerse Frankfurt do have their last trading day on 18.05.2026Die folgenden Instrumente in Boerse Frankfurt haben ihren letzten Handelstag am 18.05.2026ISIN NameAU0000248288 ADELONG... ► Artikel lesen | |
| QIAGEN | 30,290 | +0,65 % | QIAGEN N.V.: QIAGEN to advance AI-driven drug discovery with graph-based AI and curated bioinformatics knowledge with NVIDIA | Collaboration integrates NVIDIA accelerated computing and the NVIDIA BioNeMo platform with QIAGEN Digital Insights' curated biomedical knowledge bases Goal to advance how disease mechanisms... ► Artikel lesen | |
| SUMMIT THERAPEUTICS | 16,750 | -4,12 % | Cantor Fitzgerald reiterates Overweight on Summit Therapeutics stock | ||
| METAVIA | 3,800 | +32,40 % | MetaVia: Präklinische Studie zu Vanoglipel untermauert Potenzial bei Lebererkrankungen | ||
| IMMUNEERING | 5,730 | +9,77 % | Immuneering Corporation: Immuneering Reports 17.3 Months Median Overall Survival in First-Line Metastatic Pancreatic Cancer Patients Treated with Atebimetinib Plus Chemotherapy | - Detailed data from 55 patients to be shared in an oral presentation at the ASCO Annual Meeting on June 1, 2026 - - Tolerability profile consistent with prior updates: only two categories of Grade... ► Artikel lesen | |
| COGENT BIOSCIENCES | 33,720 | +3,50 % | Cogent Biosciences: Aktie legt vor wichtigen Studiendaten um 4 % zu | ||
| GEOVAX LABS | 3,900 | +41,82 % | EQS-News: GeoVax, Inc.: GeoVax Comments on Escalating Bundibugyo Ebola Outbreak and Growing Need for Flexible Biodefense Vaccine Platforms | EQS-News: GeoVax, Inc.
/ Key word(s): Science
GeoVax Comments on Escalating Bundibugyo Ebola Outbreak and Growing Need for Flexible Biodefense Vaccine Platforms 20.05.2026 / 16:06... ► Artikel lesen | |
| PRAXIS PRECISION MEDICINES | 347,16 | -0,97 % | Praxis Precision Medicines, Inc.: Praxis Precision Medicines Provides Corporate Update and Reports First Quarter 2026 Financial Results | FDA accepted the new drug application (NDA) for ulixacaltamide in Essential Tremor with a PDUFA target action date of January 29, 2027, and the NDA for relutrigine, with priority review, in SCN2A... ► Artikel lesen | |
| BIONTECH | 79,70 | +1,79 % | Ihre wichtigsten Termine: Alle Blicke auf: Fraport, Biontech, Nagarro, Freenet & Intercontinental Exchange | © Foto: UnsplashGut geplant in den Tag. Mit dem wO-Tagesausblick haben Sie die wichtigsten Termine im Blick und starten bestens vorbereitet in den neuen Handelstag.Unternehmenstermine 07:00 Uhr, Deutschland:... ► Artikel lesen | |
| HCW BIOLOGICS | 3,180 | +23,26 % | HCW Biologics Inc. - 8-K, Current Report |
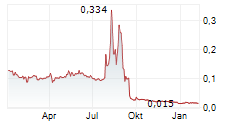
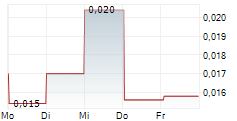
MODUS THERAPEUTICS HOLDING AB gehört der Branche Biotechnologie an. Mit einer aktuellen Kursveränderung von 0,000 (-0,70 %) liegt der Kurs bei 0,028 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu MODUS THERAPEUTICS HOLDING AB finden Sie auf Wallstreet Online.