- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 01.05. | REGENTIS BIOMATERIALS LTD. - F-1, Registration statement for certain foreign private issuers | - | SEC Filings | ||
| 16.04. | Regentis Biomaterials Ltd: Regentis Implements Strategy for Commercial Deployment of GelrinC in Europe Through Collaboration with Humanitas Hospital and Leading Cartilage Repair Expert Prof. Elizaveta Kon | 975 | ACCESS Newswire | Collaboration with leading European orthopedic center in Italy supports GelrinC clinical adoptionRenowned cartilage regeneration expert Prof. Elizaveta Kon to help lead Regentis Centers of Excellence... ► Artikel lesen | |
| 17.03. | Regentis Biomaterials Ltd: Regentis Biomaterials Develops and Patents New Solvent-Free Manufacturing Process That Increases GelrinC Production Yield by 400% | 385 | ACCESS Newswire | Breakthrough manufacturing process comes in preparation for upcoming commercial launch in Europe planned for later this yearProprietary process is already patented in several countries and is pending... ► Artikel lesen | |
| 24.02. | REGENTIS BIOMATERIALS LTD. - 20-F, Annual and transition report of foreign private issuers | 3 | SEC Filings | ||
| 24.02. | Regentis Biomaterials Ltd: Regentis Biomaterials Reports 2025 Financial Results and Provides Business Update | 735 | ACCESS Newswire | $7.4 million in cash expected to provide runway to complete site expansion process and patient enrollment as part of pivotal Phase III FDA trial for GelrinCAlready approved in Europe, GelrinC is expected... ► Artikel lesen | |
| 24.02. | REGENTIS BIOMATERIALS LTD. - 6-K, Report of foreign issuer | 2 | SEC Filings | ||
| REGENTIS BIOMATERIALS Aktie jetzt für 0€ handeln | |||||
| 24.02. | Regentis Biomaterials (RGNT) Appoints New CFO/CBO as GelrinC Nears Commercial Launch | 4 | Insider Monkey | ||
| 19.02. | Regentis Biomaterials Ltd: Regentis Biomaterials Appoints Dr. Galit Reske as Chief Medical Officer: Brings Strong Track Record of Success in Cartilage Repair | 376 | ACCESS Newswire | Dr. Reske has proven leadership in clinical development and regulatory affairs following key role in FDA approval of cartilage repair product Agili-C leading to CartiHeal's $330 million acquisition... ► Artikel lesen | |
| 17.02. | REGENTIS BIOMATERIALS LTD. - 6-K, Report of foreign issuer | 3 | SEC Filings | ||
| 17.02. | Regentis Biomaterials Ltd: Regentis Expands its European Clinical Site Network to Support its Commercial and Clinical Programs | 643 | ACCESS Newswire | With CE Mark approval, GelrinC is slated for commercial launch in Europe in 2026New clinical sites to support engagement with leading orthopedic surgeons and centers of excellence in key European markets... ► Artikel lesen | |
| 04.02. | Regentis Biomaterials appoints new CFO | 1 | Seeking Alpha | ||
| 04.02. | Regentis Biomaterials Ltd: Regentis Biomaterials Appoints Ori Gon as Chief Financial Officer and Chief Business Officer as GelrinC Progresses Towards Commercial Launch in Europe and Advances in U.S. Phase III FDA Trial | 436 | ACCESS Newswire | GelrinC, the only restorative product for knee cartilage repair, has CE Mark approval in Europe and is currently at the midpoint of a pivotal FDA Phase III trial in the U.S.Ori Gon brings substantial... ► Artikel lesen | |
| 29.01. | REGENTIS BIOMATERIALS LTD. - 6-K, Report of foreign issuer | 1 | SEC Filings | ||
| 29.01. | Regentis Biomaterials Ltd: Regentis Expanding Pivotal Phase III GelrinC Clinical Site Network: Adding Several Leading Orthopedic Centers Across the U.S. | 627 | ACCESS Newswire | New sites expected to further accelerate patient enrollment, which surpassed 50% in Phase III study, as well as support future clinical programsGelrinC is set to transform cartilage repair market with... ► Artikel lesen | |
| 20.01. | REGENTIS BIOMATERIALS LTD. - 6-K, Report of foreign issuer | 2 | SEC Filings | ||
| 20.01. | Regentis Biomaterials Ltd: Regentis' GelrinC Demonstrates Breakthrough in Regenerating Native-Like Cartilage Structure in Knee Repair on MRI Results Confirmed Using a Validated, Regulator-Accepted Method | 793 | ACCESS Newswire | 2 years after treatment, GelrinC-treated patients demonstrated layered cartilage architecture similar to native hyaline cartilage - widely regarded as the gold standard for durable joint functionThis... ► Artikel lesen | |
| 06.01. | Regentis Biomaterials Ltd: Regentis' GelrinC Establishes Long-Term Durability of Cartilage Repair Through Quantitative MOCART Evaluation | 491 | ACCESS Newswire | Marks a world-first as Regentis uses MOCART, a quantitative standard to measure regeneration of cartilage in a clinical trial, as a predefined endpointNew peer-reviewed data published in the scientific... ► Artikel lesen | |
| 06.01. | REGENTIS BIOMATERIALS LTD. - 6-K, Report of foreign issuer | 1 | SEC Filings | ||
| 18.12.25 | Regentis Biomaterials Ltd: Regentis Biomaterials Granted New U.S. Patent for its Off-the-Shelf Regenerative Cartilage Repair Product GelrinC | 477 | ACCESS Newswire | Patent protects ready to use liquid formulation of GerlinC which improves efficiency and focus for surgeons, resulting in a 10-minute procedureRobust IP portfolio includes 35 issued patents to date... ► Artikel lesen | |
| 18.12.25 | REGENTIS BIOMATERIALS LTD. - 6-K, Report of foreign issuer | 3 | SEC Filings |
1 2 Weiter >>
26 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BAYER | 38,910 | -0,82 % | Bayer: Glyphosat-Urteile lassen Anleger zittern trotz überraschend starkem Quartal | Bayer hat mit starken Quartalszahlen den Erwartungen der Analysten getrotzt, doch die Unsicherheit durch die anstehenden Glyphosat-Urteile in den USA bleibt der große Bremsklotz für die Aktie. Während... ► Artikel lesen | |
| MERCK KGAA | 126,40 | +2,97 % | BARCLAYS stuft MERCK KGAA auf 'Equal Weight' | LONDON (dpa-AFX Analyser) - Die britische Investmentbank Barclays hat die Einstufung für Merck KGaA mit einem Kursziel von 130 Euro auf "Equal Weight" belassen. Das erste Quartal des Pharma- und Chemiekonzerns... ► Artikel lesen | |
| NOVO NORDISK | 38,040 | -1,45 % | Novo Nordisk setzt große Hoffnungen auf die Wegovy-Pille | Novo Nordisk hat an der Börse schon bessere Zeiten erlebt. Nach dem jahrelangen Hype um Wegovy und Ozempic wurde der dänische Pharmakonzern zuletzt deutlich kritischer betrachtet. Sinkende Preise, stärkere... ► Artikel lesen | |
| BRIGHT MINDS BIOSCIENCES | 83,24 | 0,00 % | Bright Minds Biosciences GAAP EPS of -$1.09 for three months ended March 31 | ||
| ELI LILLY | 895,70 | -0,14 % | Dividendenbekanntmachungen (15.05.2026) | Unternehmen ISIN-Code Dividende (Währung) Dividende (EUR) A10 NETWORKS INC US0021211018 0,06 USD 0,0514 EUR AF GRUPPEN ASA NO0003078107 6,5 NOK 0,6029 EUR AGCO CORPORATION US0010841023 0... ► Artikel lesen | |
| ETON PHARMACEUTICALS | 34,990 | -0,03 % | Eton Pharmaceuticals Expands Rare Disease Portfolio Through Agreement for U.S. Commercialization Rights to IMPAVIDO (miltefosine) | IMPAVIDO is the first and only FDA-approved oral therapy for visceral, cutaneous, and mucosal leishmaniasis caused by specific Leishmania speciesLeishmaniasis is a rare but potentially life-threatening... ► Artikel lesen | |
| GENERATE BIOMEDICINES | 13,980 | 0,00 % | Generate:Biomedicines: Generate Biomedicines, Inc. Reports First Quarter 2026 Financial Results and Provides Business Update | SOLAIRIA-1 AND SOLAIRIA-2 Phase 3 clinical trials for GB-0895 (anti-TSLP) continue to advance
Clinical trial sites activated for GB-4362 (MMAE neutralizer); first... ► Artikel lesen | |
| SUNSHINE BIOPHARMA | 0,513 | -0,64 % | Sunshine Biopharma Inc.: Sunshine Biopharma Receives Canadian Regulatory Approval for Generic Amoxicillin | FORT LAUDERDALE, FL / ACCESS Newswire / May 21, 2026 / Sunshine Biopharma Inc. (NASDAQ:SBFM) (the "Company"), a leading pharmaceutical company specializing in generic and specialty prescription medications... ► Artikel lesen | |
| PFIZER | 22,295 | -0,18 % | Unterbewertet - Analysten rücken LifeScience in den Fokus! Bayer, Vidac Pharma, Novo Nordisk und Pfizer im Check | Dass Analysten den LifeScience-Bereich zuletzt wieder besser einschätzen, zeigt die dynamische Kursziel-Anpassung der DZ Bank für Bayer. Der starke Jahresstart des Pharma- und Agrarchemiekonzerns führt... ► Artikel lesen | |
| AQUESTIVE THERAPEUTICS | 4,230 | 0,00 % | Citizens reiterates Aquestive stock rating on Anaphylm progress | ||
| NOVARTIS | 131,52 | +0,38 % | Aktien Europa: Deutliche Verluste - Warnsignale vom Anleihemarkt | PARIS/LONDON/ZÜRICH (dpa-AFX) - Europas Aktienmärkte haben am Freitag einen Rücksetzer erlitten. Damit folgten sie den schwachen Vorgaben aus Asien, wo es angesichts der wieder gestiegenen Ölpreise... ► Artikel lesen | |
| ZOETIS | 68,90 | +0,03 % | ZOETIS INC: Stabilität als strategischer Wert | ||
| MERCK & CO | 99,79 | +0,08 % | BioInvent International: BioInvent Reveals Solid Data with its TNFR2 Antibody BI-1808 and KEYTRUDA in Advanced Ovarian Cancer at ASCO | BioInvent International (STO:BINV)24% confirmed ORR (including complete response) in heavily pretreated patients (n=25) with advanced ovarian cancer who received BI-1808 and KEYTRUDA - multiplying by... ► Artikel lesen | |
| SANOFI | 77,36 | -0,08 % | Entwicklung von neuen Medikamenten: Wie die Durchführung klinischer Studien bei Sanofi koordiniert wird | Frankfurt/Main (ots) - Klinische Studien sind mit der wichtigste Schritt bei der Entwicklung von Medikamenten. Mit ihnen werden neue Wirkstoffe unter realen Bedingungen getestet. Die genau geplanten... ► Artikel lesen | |
| SCHOTT PHARMA | 16,660 | +0,36 % | DZ BANK stuft Schott Pharma auf 'Kaufen' | FRANKFURT (dpa-AFX Analyser) - Die DZ Bank hat den fairen Wert für die Aktien von Schott Pharma von 22 auf 21 Euro gesenkt, aber die Einstufung auf "Kaufen" belassen. Das erste Geschäftshalbjahr sei... ► Artikel lesen |
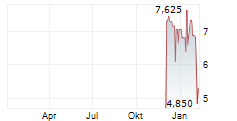
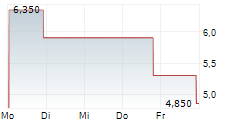
REGENTIS BIOMATERIALS LTD gehört der Branche Pharma an. Die relative Kursveränderung beträgt aktuell +0,95 % bei einem Aktienkurs von 2,120 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu REGENTIS BIOMATERIALS LTD finden Sie auf Wallstreet Online.