- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| ETON PHARMACEUTICALS Aktie jetzt für 0€ handeln | |||||
| 23.05. | H.C. Wainwright Remains Bullish on Eton Pharmaceuticals' (ETON) Growth Strategy | 1 | Insider Monkey | ||
| 21.05. | H.C. Wainwright reiterates Eton Pharmaceuticals stock rating at buy | 20 | Investing.com | ||
| 19.05. | Eton Pharmaceuticals acquires U.S. rights to leishmaniasis drug | 1 | Investing.com | ||
| 19.05. | Eton Pharmaceuticals, Inc. - 8-K, Current Report | 2 | SEC Filings | ||
| 19.05. | Eton Pharmaceuticals Expands Rare Disease Portfolio Through Agreement for U.S. Commercialization Rights to IMPAVIDO (miltefosine) | 327 | GlobeNewswire (Europe) | IMPAVIDO is the first and only FDA-approved oral therapy for visceral, cutaneous, and mucosal leishmaniasis caused by specific Leishmania speciesLeishmaniasis is a rare but potentially life-threatening... ► Artikel lesen | |
| 18.05. | H.C. Wainwright hebt Kursziel für Eton Pharmaceuticals auf 57 US-Dollar an | 1 | Investing.com Deutsch | ||
| 18.05. | H.C. Wainwright raises Eton Pharmaceuticals stock price target to $57 | 1 | Investing.com | ||
| 14.05. | Full Transcript: Eton Pharmaceuticals Q1 2026 Earnings Call | 1 | Benzinga.com | ||
| 14.05. | Eton Pharmaceuticals, Inc. (ETON) Misses Q1 Earnings Estimates | 1 | Zacks | ||
| 14.05. | Eton Pharmaceuticals, Inc. - 10-Q, Quarterly Report | 1 | SEC Filings | ||
| 04.05. | Eton Pharmaceuticals (ETON) Surges 19.8%: Is This an Indication of Further Gains? | 3 | Zacks | ||
| 01.05. | Eton Pharmaceuticals relaunches Hemangeol with specialty pharmacy | 3 | Investing.com | ||
| 01.05. | Eton Pharmaceuticals relaunches HEMANGEOL (propranolol) Oral Solution with Eton Cares and Exclusive Specialty Pharmacy Distribution | 286 | GlobeNewswire (Europe) | HEMANGEOL is now available exclusively through Anovo Specialty Pharmacy to streamline access and therapy initiationEton has integrated full Eton Cares patient support, including $0 co-pay for eligible... ► Artikel lesen | |
| 27.04. | Eton Pharmaceuticals doses first patient in Wilson disease study | 2 | Investing.com | ||
| 27.04. | Eton Pharmaceuticals Announces Initiation of Clinical Study for Product Candidate ET-700 | 349 | GlobeNewswire (Europe) | DEER PARK, Ill., April 27, 2026 (GLOBE NEWSWIRE) -- Eton Pharmaceuticals, Inc ("Eton" or "the Company") (Nasdaq: ETON), an innovative pharmaceutical company focused on developing and commercializing... ► Artikel lesen | |
| 27.04. | Eton Pharmaceuticals, Inc. - 8-K, Current Report | - | SEC Filings | ||
| 24.04. | Eton Pharmaceuticals (ETON) Appoints Judith M. Matthews as New CFO | 1 | Insider Monkey | ||
| 16.04. | Eton Pharma drops after CFO succession plan | 1 | Seeking Alpha | ||
| 16.04. | Eton Pharmaceuticals Announces CFO Succession Plan | 271 | GlobeNewswire (Europe) | DEER PARK, Ill., April 16, 2026 (GLOBE NEWSWIRE) -- Eton Pharmaceuticals, Inc. ("Eton" or the "Company") (Nasdaq: ETON), an innovative pharmaceutical company focused on developing and commercializing... ► Artikel lesen | |
| 15.04. | Eton Pharmaceuticals, Inc. - 8-K, Current Report | - | SEC Filings |
1 2 3 Weiter >>
41 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BAYER | 35,580 | -0,31 % | Bayer-Aktie bricht ein: Rechtsrisiken lassen Anleger zittern | Erneuter Kursrutsch bei Bayer sorgt für Unruhe an den Märkten: Die Aktie fällt auf ein neues Sechsmonatstief. Wachsende Zweifel an einem günstigen Ausgang der Glyphosat-Klagen setzen das Papier zusätzlich... ► Artikel lesen | |
| NOVO NORDISK | 36,000 | +0,85 % | GOLDMAN SACHS stuft NOVO NORDISK auf 'Neutral' | NEW YORK (dpa-AFX Analyser) - Die US-Investmentbank Goldman Sachs hat das Kursziel für Novo Nordisk von 263 auf 305 dänische Kronen angehoben, aber die Einstufung auf "Neutral" belassen. Das Abnehmmittel... ► Artikel lesen | |
| PFIZER | 22,150 | -0,29 % | Rendite-Chance Krebsmarkt: Mit BioNTech, Vidac Pharma und Pfizer den nächsten Mrd.-Trigger erwischen | Die Alterung der Gesellschaft lässt die Gesundheitsausgaben rasant steigen. Im Krebssegment zeichnet sich ein Billionenmarkt ab, denn bis 2040 werden jährlich über 30 Mio. Neuerkrankungen erwartet.... ► Artikel lesen | |
| NOVARTIS | 127,22 | +0,39 % | JEFFERIES stuft NOVARTIS AG auf 'Hold' | NEW YORK (dpa-AFX Analyser) - Das Analysehaus Jefferies hat die Einstufung für Novartis mit einem Kursziel von 110 Franken auf "Hold" belassen. Der Entwicklungschef habe sich sehr positiv zum Sicherheitsprofil... ► Artikel lesen | |
| GILEAD SCIENCES | 107,38 | -3,37 % | Neue CAR-T-Erfolge bei Autoimmunerkrankungen: Warum Lösungen für Zelltherapien vor dem nächsten Wachstumsschub stehen könnten | ||
| AURORA CANNABIS | 2,994 | +1,08 % | Aurora Deepens its Impact for Veterans Across Canada | ||
| ABBVIE | 191,40 | -1,03 % | Drei Qualitätswerte im Fokus: Was der "Quant Growth And Income"-Ansatz jetzt über Microsoft, Visa und AbbVie verrät | Microsoft, Visa und AbbVie stehen im Zentrum eines quantitativen Wachstums- und Einkommensansatzes, den Seeking Alpha aktuell transparent macht. Der Einblick in das "Quant Growth And Income"-Portfolio... ► Artikel lesen | |
| ROCHE | 348,07 | -0,49 % | JEFFERIES stuft ROCHE HOLDINGS AG auf 'Underperform' | NEW YORK (dpa-AFX Analyser) - Das Analysehaus Jefferies hat die Einstufung für Roche mit einem Kursziel von 230 Franken auf "Underperform" belassen. Das Risiko für die mittelfristigen Ziele der Schweizer... ► Artikel lesen | |
| CANOPY GROWTH | 0,920 | +3,51 % | Canopy Growth Just Made a Big Acquisition: Game-Changer -- or Another Misstep? | ||
| ELI LILLY | 992,00 | -0,42 % | ELI LILLY ist zuletzt auf ein Allzeithoch gestiegen | Der zentrale Auslöser war die neue Erstattungsvereinbarung mit CVS Caremark. Der Pharmacy-Benefit-Manager nimmt die GLP-1-Pille Foundayo ab 1. Juni und die Abnehm-Injektion Zepbound ab 1. Oktober in... ► Artikel lesen | |
| MERCK & CO | 102,42 | -1,18 % | Rejuvenate Bio Raises $6 Million And Partners With Merck Animal Health To Advance Gene Therapy Programs | ||
| BRISTOL-MYERS SQUIBB | 48,045 | -0,34 % | Bristol-Myers Squibb: Wird dieser Pharmakonzern massiv unterschätzt? | Die Aktie von Bristol Myers Squibb steht seit Jahren unter dem Eindruck auslaufender Blockbuster-Medikamente und drohender Patentverluste. Doch die jüngsten Geschäftszahlen zeigen ein Unternehmen, dessen... ► Artikel lesen | |
| VERTEX PHARMACEUTICALS | 385,15 | +0,30 % | Vertex Pharmaceuticals Incorporated: Vertex Presents New Data on ALYFTREK at European Cystic Fibrosis Conference | - ALYFTREK Phase 3 data on children with cystic fibrosis ages 2 to 5 with vanzacaftor/tezacaftor/deutivacaftor-responsive genotypes including F/F and F/MF shows 65% reached sweat chloride levels... ► Artikel lesen | |
| EYEPOINT | 10,000 | +1,52 % | EyePoint, Inc.: EyePoint Announces Third Consecutive Positive DSMC Recommendation for Phase 3 Wet AMD Trials for DURAVYU, Building Confidence Ahead of Mid-2026 Topline Data | - LUGANO topline data on track for mid-2026, with LUCIA readout to follow shortly after - - Continued favorable safety profile observed in masked Phase 3 DURAVYU data, consistent with four previously... ► Artikel lesen | |
| INCYTE | 87,02 | -0,30 % | XETR NEW INSTRUMENTS AVAILABLE ON XETRA - 04.06.2026 | Folgende Wertpapiere werden in den Handel an folgenden Handelsplätzen (MIC Code) im Quotation Board an der Frankfurter Wertpapierbörse (FWB) aufgenommen. Die Notierung an anderen Handelsplätzen im Quotation... ► Artikel lesen |
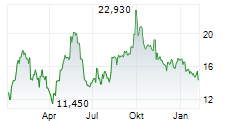
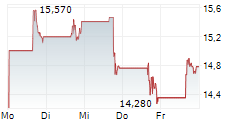
ETON PHARMACEUTICALS INC gehört der Branche Pharma an. Mit einer aktuellen Kursveränderung von +0,140 (+0,47 %) liegt der Kurs bei 30,090 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu ETON PHARMACEUTICALS INC finden Sie auf Wallstreet Online.