- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| SCANCELL Aktie jetzt für 0€ handeln | |||||
| 28.04. | Scancell Holdings Plc: Scancell receives FDA Fast Track Designation for iSCIB1+ in advanced melanoma and provides data update from its SCOPE Phase 2 study | 135 | GlobeNewswire (Europe) | Potent and durable efficacy with iSCIB1+ of 77% progression free survival (PFS) at 20 months, in combination with ipilimumab and nivolumab, demonstrated in the Phase 2 SCOPE trial; more PFS and additional... ► Artikel lesen | |
| 28.04. | Scancell Holdings - FDA Fast Track Designation for iSCIB1+ | - | RNS | ||
| 02.04. | IN BRIEF: Scancell names David Schilansky as interim finance chief | 3 | Alliance News | ||
| 02.04. | Scancell Holdings - Scancell announces CFO transition | - | RNS | ||
| 29.01. | Scancell Holdings plc GAAP EPS of -£0.64p | 1 | Seeking Alpha | ||
| 29.01. | Scancell Holdings Plc: Interim Results for the six months ended 31 October 2025 | 405 | GlobeNewswire (Europe) | OXFORD, United Kingdom, Jan. 29, 2026 (GLOBE NEWSWIRE) -- Scancell Holdings plc (AIM: SCLP), the developer of ImmunoBody and Moditope active immunotherapies to treat cancer, today announces a business... ► Artikel lesen | |
| 29.01. | Scancell Holdings - Half-year Financial Report | - | RNS | ||
| 26.01. | EARNINGS AND TRADING: Scancell gets FDA clearance for phase 3 trial | 1 | Alliance News | ||
| 26.01. | FDA clears Scancell's application for phase three trial | 2 | Sharecast | ||
| 26.01. | Scancell Holdings Plc: Scancell announces FDA clearance of IND application for global Phase 3 trial of iSCIB1+ in advanced melanoma | 274 | GlobeNewswire (Europe) | Unlocks path towards registrational Phase 3 trial planned to start in 2026 Data from Phase 2 SCOPE trial show iSCIB1+ has potential to redefine standard of care (SoC) iSCIB1+ shows an interim 24%-point... ► Artikel lesen | |
| 26.01. | Scancell Holdings - FDA clearance of IND application for iSCIB1+ | - | RNS | ||
| 22.01. | Scancell Holdings - Notice of Results | - | RNS | ||
| 09.12.25 | Scancell reports further data from DNA cancer immunotherapy trial | 2 | Sharecast | ||
| 09.12.25 | Scancell Holdings Plc: Scancell updated Phase 2 data shows continued improvement in progression free survival with iSCIB1+ in patients with first line advanced melanoma | 1 | GlobeNewswire (USA) | ||
| 09.12.25 | Scancell Holdings - Continued improvement in PFS with iSCIB1+ | 1 | RNS | ||
| 07.11.25 | Scancell Holdings Plc: Research Update | 283 | GlobeNewswire (Europe) | Scancell Holdings plc Scancell holds oral presentation of positive Phase 2 data on Immunobody® iSCIB1+ in late-stage melanoma at SITC 2025 Data from SCOPE trial show a potential new benchmark in... ► Artikel lesen | |
| 11.09.25 | Scancell Holdings Plc: Scancell reports Business Update and Financial Results for the Year Ended 30 April 2025 | 499 | GlobeNewswire (Europe) | NOTTINGHAM, United Kingdom, Sept. 11, 2025 (GLOBE NEWSWIRE) -- Scancell Holdings plc (AIM: SCLP), the developer of ImmunoBody® and Moditope® active immunotherapies to treat cancer, today announces... ► Artikel lesen | |
| 22.07.25 | Scancell Holdings Plc: Scancell reports Phase 2 data showing strongly improved outcomes in Late-Stage Melanoma with its Immunobody iSCIB1+ | 254 | GlobeNewswire (Europe) | SCOPE trial of SCIB1/iSCIB1+ plus standard of care shows excellent results encompassing efficacy, durability, immune responses and safety Overall response rate for iSCIB1+ was 69% for target HLA type... ► Artikel lesen |
18 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 78,05 | -1,51 % | Vorsicht bei BioNTech! Novo Nordisk gibt Gas! Kaufchance bei Vidac Pharma Aktie! | Kaufstimmung bei Novo Nordisk. Die Aktie des dänischen Pharmagiganten scheint den seit Sommer 2024 begonnenen Abwärtstrend zu verlassen. Anleger reagieren auf positive Meldungen. Bei Vidac Pharma ist... ► Artikel lesen | |
| EVOTEC | 5,160 | -2,73 % | EQS-NVR: Evotec SE: Veröffentlichung der Gesamtzahl der Stimmrechte nach § 41 WpHG mit dem Ziel der europaweiten Verbreitung | EQS Gesamtstimmrechtsmitteilung: Evotec SE
/ Veröffentlichung der Gesamtzahl der Stimmrechte
Evotec SE: Veröffentlichung der Gesamtzahl der Stimmrechte nach § 41 WpHG mit dem... ► Artikel lesen | |
| MEDIGENE | 0,027 | -6,21 % | PTA-Adhoc: Medigene AG: Widerruf der Zulassung der Aktien der Medigene AG zum Handel im regulierten Markt | DJ PTA-Adhoc: Medigene AG: Widerruf der Zulassung der Aktien der Medigene AG zum Handel im regulierten Markt
Veröffentlichung von Insiderinformationen gemäß Artikel 17 MAR Schlagwort(e): Sonstiges... ► Artikel lesen | |
| QIAGEN | 31,115 | +0,97 % | DEUTSCHE BANK RESEARCH stuft QIAGEN NV auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat die Einstufung für Qiagen mit einem Kursziel von 43 US-Dollar auf "Buy" belassen. Das Interesse der Investoren habe vor allem dem Tuberkulosetest... ► Artikel lesen | |
| PAION | 0,065 | -21,21 % | XFRA MISTRADE ANTRAG IN ISIN DE000A3E5EG5 WURDE STATTGEGEBEN | Dem Mistrade-Antrag wurde stattgegeben. Folgende Geschaefte in der ISIN DE000A3E5EG5, Wertpapier-Name: PAION AG INH O.N., wurden aufgehoben:ISIN Datum Zeit Volumen PreisDE000A3E5EG5 02.02.2026 08:13:40 300 0... ► Artikel lesen | |
| VALNEVA | 2,500 | -0,56 % | Original-Research: Valneva SE (von First Berlin Equity Research GmbH): Buy | Original-Research: Valneva SE - von First Berlin Equity Research GmbH
28.05.2026 / 17:08 CET/CEST
Veröffentlichung einer Research, übermittelt durch EQS News - ein Service der EQS Group.
Für den... ► Artikel lesen | |
| EPIGENOMICS | 0,870 | -1,14 % | PTA-HV: Epigenomics AG: Einladung zur ordentlichen Hauptversammlung | DJ PTA-HV: Epigenomics AG: Einladung zur ordentlichen Hauptversammlung
Hauptversammlung gemäß -- 121 Abs. 4a AktG
Epigenomics AG: Einladung zur ordentlichen Hauptversammlung
Heidelberg... ► Artikel lesen | |
| NOVAVAX | 9,230 | +2,10 % | Novavax, Inc. (NVAX) Is a Trending Stock: Facts to Know Before Betting on It | ||
| BIOFRONTERA | 2,530 | +3,69 % | PTA-News: Biofrontera AG: Biofrontera berichtet über die Ergebnisse des ersten Quartals 2026 | DJ PTA-News: Biofrontera AG: BIOFRONTERA BERICHTET ÜBER DIE ERGEBNISSE DES ERSTEN QUARTALS 2026
Unternehmensmitteilung für den Kapitalmarkt
Biofrontera AG: BIOFRONTERA BERICHTET ÜBER... ► Artikel lesen | |
| HEIDELBERG PHARMA | 2,780 | +1,46 % | EQS-HV: Heidelberg Pharma AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in Ladenburg mit dem Ziel der europaweiten Verbreitung gemäß §121 AktG | EQS-News: Heidelberg Pharma AG
/ Bekanntmachung der Einberufung zur Hauptversammlung
Heidelberg Pharma AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in... ► Artikel lesen | |
| 4SC | 0,100 | -23,37 % | XFRA DELETION OF INSTRUMENTS FROM BOERSE FRANKFURT - 16.12.2025 | The following instruments on Boerse Frankfurt do have their last trading day on 16.12.2025Die folgenden Instrumente in Boerse Frankfurt haben ihren letzten Handelstag am 16.12.2025ISIN NameDE000A3E5C40 4SC... ► Artikel lesen | |
| BRAIN BIOTECH | 2,960 | -1,33 % | EQS-News: BRAIN Biotech AG: BRAIN Biotech veröffentlicht Halbjahreszahlen für das Geschäftsjahr 2025/26 und bestätigt Gesamtjahresprognose | EQS-News: BRAIN Biotech AG
/ Schlagwort(e): Halbjahresergebnis/Halbjahresbericht
BRAIN Biotech veröffentlicht Halbjahreszahlen für das Geschäftsjahr 2025/26 und bestätigt Gesamtjahresprognose... ► Artikel lesen | |
| CO.DON | 0,014 | -25,00 % | EQS-AFR: CO.DON AG i.l.: Vorabbekanntmachung über die Veröffentlichung von Finanzberichten gemäß §§ 114, 115, 117 WpHG | EQS Vorabbekanntmachung Finanzberichte: CO.DON AG i.l.
/ Vorabbekanntmachung über die Veröffentlichung von Rechnungslegungsberichten
CO.DON AG i.l.: Vorabbekanntmachung über die... ► Artikel lesen | |
| INOVIO PHARMACEUTICALS | 1,067 | -3,44 % | H.C. Wainwright senkt Kursziel für Inovio wegen höherer Aktienanzahl | ||
| VIKING THERAPEUTICS | 27,100 | +0,74 % | Viking Therapeutics, Inc. (VKTX) Exceeds Market Returns: Some Facts to Consider |
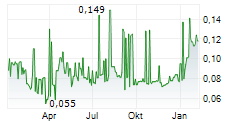
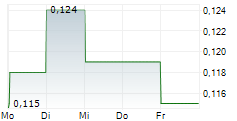
Das Unternehmen SCANCELL HOLDINGS PLC kann der Branche Biotechnologie zugeordnet werden. Der aktuelle Kurs liegt bei 0,248 Euro und damit -13,89 % im Minus.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu SCANCELL HOLDINGS PLC finden Sie auf Wallstreet Online.