- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 19.05. | Egetis Therapeutics: Egetis to Present Healthcare Resource Drivers and Cost in MCT8 Deficiency at ISPOR 2026 | 436 | ACCESS Newswire | STOCKHOLM, SE / ACCESS Newswire / May 19, 2026 / Egetis Therapeutics AB (publ) ("Egetis" or the "Company") (STO:EGTX)(NASDAQ Stockholm:EGTX) today announced its participation in the annual conference... ► Artikel lesen | |
| 07.05. | Egetis Therapeutics to Participate in Leading International Medical Conferences in 2026 | 393 | ACCESS Newswire | STOCKHOLM, SE / ACCESS Newswire / May 7, 2026 / Egetis Therapeutics AB (publ) ("Egetis" or the "Company") (Nasdaq Stockholm:EGTX) today announced that the Company is planning to participate in several... ► Artikel lesen | |
| 05.05. | Egetis Therapeutics: Egetis Announces Grant of Patent for MCT8 Deficiency Composition in The U.S. | 377 | ACCESS Newswire | STOCKHOLM, SE / ACCESS Newswire / May 5, 2026 / Egetis Therapeutics AB (publ) ("Egetis" or the "Company") (NASDAQ Stockholm:EGTX) today announced that the United States Patent and Trademark Office (USPTO)... ► Artikel lesen | |
| 30.04. | Egetis Therapeutics to Participate in Pediatric Endocrine Society Annual Meeting 2026 | 230 | ACCESS Newswire | STOCKHOLM, SE / ACCESS Newswire / April 30, 2026 / Egetis Therapeutics (STO:EGTX) - Egetis Therapeutics AB (publ) ("Egetis" or the "Company") (NASDAQ Stockholm:EGTX), today announced its participation... ► Artikel lesen | |
| 29.04. | Egetis Therapeutics AB: Interim report Q1 2026 | 242 | GlobeNewswire (Europe) | Egetis Announced FDA Acceptance and Priority Review of NDA for Emcitate® (tiratricol) for MCT8 Deficiency · Egetis completed the U.S. New Drug Application (NDA) for Emcitate® in January· ... ► Artikel lesen | |
| 21.04. | Egetis Therapeutics AB: Egetis Therapeutics has successfully carried out a directed share issue amounting to SEK 350 million (approximately USD 38 million) | 139 | GlobeNewswire (Europe) | NOT FOR PUBLICATION, DISTRIBUTION OR RELEASE, DIRECTLY OR INDIRECTLY, IN WHOLE OR IN PART, IN OR INTO THE UNITED STATES OF AMERICA, AUSTRALIA, CANADA, HONG KONG, ISRAEL, JAPAN, NEW ZEALAND, SOUTH AFRICA... ► Artikel lesen | |
| 14.04. | Egetis Therapeutics AB: Bulletin from Egetis Therapeutics' Annual General Meeting 2026 | 301 | GlobeNewswire (Europe) | Stockholm, Sweden, April 14, 2026. Egetis Therapeutics AB (publ) (STO: EGTX) today announced that the Annual General Meeting (AGM) has been held on April 14, 2026, at which the submitted proposals were... ► Artikel lesen | |
| 09.04. | Egetis Therapeutics: Egetis Appoints Tiago Nunes as Chief Medical Officer | 631 | ACCESS Newswire | STOCKHOLM, SE / ACCESS Newswire / April 9, 2026 / Egetis Therapeutics AB (publ) ("Egetis" or the "Company") (STO:EGTX), today announced the appointment of Tiago Nunes, MD, PhD, as Chief Medical Officer... ► Artikel lesen | |
| EGETIS THERAPEUTICS Aktie jetzt für 0€ handeln | |||||
| 27.03. | Egetis Therapeutics: Egetis Announces FDA Acceptance and Priority Review of NDA for Emcitate for MCT8 Deficiency | 588 | ACCESS Newswire | NDA submission for Emcitate successfully validated by the FDAPriority Review granted and PDUFA target action date set to September 28, 2026Emcitate (tiratricol) is eligible to receive a Priority Review... ► Artikel lesen | |
| 09.03. | Egetis Therapeutics: Egetis Receives Notice of Allowance for MCT8 Deficiency Composition Patent in the U.S. | 417 | ACCESS Newswire | STOCKHOLM, SE / ACCESS Newswire / March 9, 2026 / Egetis Therapeutics AB (publ) ("Egetis" or the "Company") (NASDAQ Stockholm:EGTX) today announced that the United States Patent and Trademark Office... ► Artikel lesen | |
| 02.03. | Egetis Therapeutics Launches Educational Websites on MCT8 Deficiency for Physicians and Caregivers | 348 | ACCESS Newswire | STOCKHOLM, SWEDEN / ACCESS Newswire / March 2, 2026 / Egetis Therapeutics AB (publ) ("Egetis" or the "Company") (NASDAQ Stockholm:EGTX), today announced the launch of two updated educational websites... ► Artikel lesen | |
| 26.02. | Egetis Therapeutics AB: Year-End Report January-December 2025 | 272 | GlobeNewswire (Europe) | Egetis submitted the U.S. NDA for Emcitate® (tiratricol) for treatment of MCT8 deficiency· Emcitate® revenue during 2025 amounted to MSEK 62.3, a 40 % increase in constant exchange rates from... ► Artikel lesen | |
| 16.02. | Egetis Therapeutics AB: Egetis provides update on progress with the development of Emcitate (tiratricol) for MCT8 deficiency in Japan | 265 | GlobeNewswire (Europe) | Stockholm, Sweden, February 16, 2026. Egetis Therapeutics AB (publ) ("Egetis" or the "Company") (NASDAQ Stockholm: EGTX), today announced that its Japanese partner Fujimoto Pharmaceuticals, who has... ► Artikel lesen | |
| 04.02. | Egetis Therapeutics AB: FDA's rare pediatric disease priority review program extended until 2029 | 296 | GlobeNewswire (Europe) | Stockholm, Sweden, February 4, 2026. Egetis Therapeutics AB (publ) ("Egetis" or the "Company") (NASDAQ Stockholm: EGTX), today noted that the U.S. Food and Drug Administration's (FDA's) Rare Pediatric... ► Artikel lesen | |
| 29.01. | Egetis Therapeutics AB: Egetis Completes the U.S. Rolling NDA Submission for Emcitate (tiratricol) for Treatment of MCT8 Deficiency | 228 | GlobeNewswire (Europe) | Priority Review and Rare Pediatric Disease Priority Review Voucher requestedAnticipated regulatory decision in September 2026Emcitate® (tiratricol) U.S. launch expected in Q4 2026, if approvedStockholm... ► Artikel lesen | |
| 19.12.25 | Egetis Therapeutics AB: Egetis Therapeutics initiates New Drug Application in the USA for Emcitate (tiratricol) for MCT8 deficiency | 307 | GlobeNewswire (Europe) | Stockholm, Sweden, December 19, 2025. Egetis Therapeutics AB (publ) ("Egetis" or the "Company") (NASDAQ Stockholm: EGTX), today announced that the Company has initiated a rolling New Drug Application... ► Artikel lesen | |
| 10.12.25 | Egetis Therapeutics AB: Egetis and Er-Kim Expand Partnership to Broaden Access to Emcitate Across Central, Eastern, and Southeastern Europe | 224 | GlobeNewswire (Europe) | Stockholm, Sweden, December 10, 2025. Egetis Therapeutics AB (publ) ("Egetis" or the "Company") (NASDAQ Stockholm: EGTX), today announced that the Company, through its wholly-owned subsidiary Rare Thyroid... ► Artikel lesen | |
| 25.11.25 | Egetis Therapeutics AB: Interim report Q3 2025 | 247 | GlobeNewswire (Europe) | Egetis to commence a rolling NDA submission in December as granted by the FDA for Emcitate® (tiratricol)· Egetis received FDA Breakthrough Therapy Designation for Emcitate® (tiratricol) for... ► Artikel lesen | |
| 14.11.25 | Egetis Therapeutics AB: Egetis announces positive results from the ReTRIACt study of Emcitate (tiratricol) in MCT8 deficiency | 209 | GlobeNewswire (Europe) | Positive topline results demonstrate a statistically significant (p=0.034) difference in the rate of change in serum T3 in patients randomized to withdrawal (placebo) vs. in patients continuing therapy... ► Artikel lesen | |
| 23.10.25 | Egetis Therapeutics AB: Egetis granted a rolling NDA review by FDA for Emcitate (tiratricol) based on currently available clinical data | 330 | GlobeNewswire (Europe) | The outcomes of a successful pre-NDA meeting with the FDA are:
As agreed with the FDA, the rolling NDA submission will commence in December 2025, targeting a complete NDA submission in early 2026 and... ► Artikel lesen |
1 2 Weiter >>
24 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BAYER | 33,350 | -5,07 % | Bayer-Aktie: Das könnte brandgefährlich werden | Die Bayer-Aktie pendelte in den letzten drei Monaten in einem Seitwärtstrend zwischen 36 und 41 € auf und ab. Doch nun könnte eine Nachricht die Aktie aus diesem Trend nach unten reißen. Wie sollten... ► Artikel lesen | |
| SANOFI | 73,91 | -0,36 % | DEUTSCHE BANK RESEARCH stuft SANOFI auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat die Einstufung für Sanofi mit einem Kursziel von 100 Euro auf "Buy" belassen. In den Gesprächen mit den Franzosen sei es vor allem um den Umbruch... ► Artikel lesen | |
| GSK | 21,130 | -0,56 % | GSK erzielt teilweisen Therapieerfolg mit Hepatitis-B-Wirkstoff | DJ GSK erzielt teilweisen Therapieerfolg mit Hepatitis-B-Wirkstoff
Von Adria Calatayud
DOW JONES--GSK hat mit einem Medikamentenkandidaten gegen Hepatitis B einen teilweisen Therapieerfolg... ► Artikel lesen | |
| ASTRAZENECA | 155,00 | +0,32 % | Astrazeneca liefert FDA neue Daten für US-Zulassung von Camizestrant nach | DJ Astrazeneca liefert FDA neue Daten für US-Zulassung von Camizestrant nach
Von Adria Calatayud
DOW JONES--Die US-Arzneimittelbehörde FDA verschiebt ihre Entscheidung über die Zulassung eines... ► Artikel lesen | |
| TILRAY BRANDS | 4,730 | +0,11 % | Tilray Brands, Inc.: BrewDog verwandelt die diesjährige Fußball-WM in ein landesweites Sommerfest rund um Bier, Bars und große Spielmoment | BrewDog startet die Multichannel-Kampagne "Underdog" im Einzelhandel, in Bars und in digitalen Medien - einschließlich des landesweiten Gewinnspiels "Win a Year of Beer" und öffentlicher Public-Viewing-Events... ► Artikel lesen | |
| DERMAPHARM | 47,500 | +0,74 % | ANALYSE-FLASH: Jefferies hebt Ziel für Dermapharm auf 44 Euro - 'Hold' | NEW YORK (dpa-AFX Broker) - Das Analysehaus Jefferies hat das Kursziel für Dermapharm von 34 auf 44 Euro angehoben, aber die Einstufung auf "Hold" belassen. Die Profitabilität des Spezialisten für... ► Artikel lesen | |
| ZOETIS | 66,74 | +0,09 % | Argus downgrades Zoetis stock rating on competition, margin pressure | ||
| DOCMORRIS | 7,815 | -0,19 % | Stiftung Warentest testet Online-Apotheken: DocMorris ist Testsieger | Berlin (ots) - Die Stiftung Warentest hat neun Online-Apotheken getestet und berichtet darüber in der test Ausgabe 6/26. Auf Platz 1 steht DocMorris, dicht gefolgt vom Schwesterunternehmen medpex auf... ► Artikel lesen | |
| SCHOTT PHARMA | 17,620 | +2,20 % | SCHOTT PHARMA AG & CO KGAA: Stabilität als Signal | ||
| QUANTUM BIOPHARMA | 4,512 | -0,44 % | Quantum BioPharma Provides Corporate Update | THIS NEWS RELEASE IS INTENDED FOR DISTRIBUTION IN CANADA ONLY AND IS NOT INTENDED FOR DISTRIBUTION TO UNITED STATES NEWSWIRE SERVICES OR DISSEMINATION IN THE UNITED STATES. TORONTO, June 01, 2026... ► Artikel lesen | |
| CSPC PHARMA | 0,840 | +1,18 % | CSPC PHARMA (01093): VOLUNTARY ANNOUNCEMENT - FIRST MRNA-LNP-BASED DUAL-TARGET CAR-T CELL INJECTION (SYS6063 INJECTION) OBTAINS CLINICAL TRIAL APPROVAL ... | ||
| NEKTAR THERAPEUTICS | 54,68 | -0,47 % | Russell 3000 Shuffle: Ocugen, Nektar slated to join among healthcare stocks; Nano-X to drop off | ||
| TONIX PHARMACEUTICALS | 10,745 | +1,66 % | Tonix Pharmaceuticals Holding Corp.: Tonix Pharmaceuticals Presented Retrospective U.S. Real-World Claims Analysis Characterizing Patients with Fibromyalgia at The Professional Society for Health Economics and Outcomes Research (ISPOR) 2026 ... | Real-world analysis of three years of closed claims data from Symphony Health focused on the third year of the study (2023-2024), comprised of more than 261,000 U.S. adults with fibromyalgia Data... ► Artikel lesen | |
| FILANA THERAPEUTICS | 1,276 | -1,09 % | Filana Therapeutics, Inc.: Filana Therapeutics Reports Q1 2026 Financial Results and Business Update | AUSTIN, Texas, May 07, 2026 (GLOBE NEWSWIRE) -- Filana Therapeutics, Inc. (NASDAQ: FLNA, "Filana Therapeutics", the "Company"), a biotechnology company currently focused on developing therapies for... ► Artikel lesen | |
| CANTOURAGE GROUP | 6,520 | 0,00 % | Original-Research: Cantourage Group SE (von NuWays AG): BUY | Original-Research: Cantourage Group SE - from NuWays AG
12.05.2026 / 09:00 CET/CEST
Dissemination of a Research, transmitted by EQS News - a service of EQS Group.
The issuer is solely responsible... ► Artikel lesen |
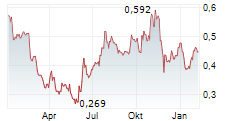
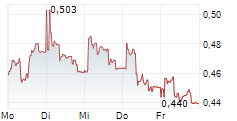
Das Unternehmen EGETIS THERAPEUTICS AB kann der Branche Pharma zugeordnet werden. Mit einer aktuellen Kursveränderung von +0,001 (+0,19 %) liegt der Kurs bei 0,539 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu EGETIS THERAPEUTICS AB finden Sie auf Wallstreet Online.