- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| Fr | Theriva Biologics, Inc. - 8-K, Current Report | 4 | SEC Filings | ||
| 05.05. | Theriva Biologics GAAP EPS of -$0.05 in-line, revenue of $0.3M | 2 | Seeking Alpha | ||
| 05.05. | Theriva Biologics, Inc.: Theriva Biologics Reports First Quarter 2026 Operational Highlights and Financial Results | 363 | GlobeNewswire (Europe) | - Alignment with the FDA on the Phase 3 trial design of VCN-01 (zabilugene almadenorepvec) for treatment of metastatic pancreatic ductal adenocarcinoma (PDAC) -
- Additional data from VIRAGE Phase... ► Artikel lesen | |
| 05.05. | Theriva Biologics, Inc. - 10-Q, Quarterly Report | 1 | SEC Filings | ||
| 05.05. | Theriva Biologics, Inc. - 8-K, Current Report | 2 | SEC Filings | ||
| 20.04. | Theriva Biologics: Aktie springt nach vielversprechenden Daten aus Krebsstudie an | 5 | Investing.com Deutsch | ||
| 17.04. | Theriva to present VCN-01 pancreatic cancer trial data at AACR | 1 | Investing.com | ||
| THERIVA BIOLOGICS Aktie jetzt für 0€ handeln | |||||
| 17.04. | Theriva präsentiert auf AACR-Jahrestagung neue Studiendaten zu Bauchspeicheldrüsenkrebs | 1 | Investing.com Deutsch | ||
| 17.04. | Theriva Biologics, Inc.: Theriva Biologics Announces Upcoming Presentation of Additional Data from the VIRAGE Phase 2b Clinical Trial of VCN-01 in Metastatic Pancreatic Cancer at AACR 2026 Annual Meeting | 522 | GlobeNewswire (Europe) | - Tumor reponse, biomarker, and subgroup analyses from the VIRAGE Phase 2b clinical trial support a VCN-01 immune-mediated mode of action and demonstrate improved outcomes in VCN-01 treated patients... ► Artikel lesen | |
| 09.04. | Mangelnde Beschlussfähigkeit: Theriva Biologics muss Hauptversammlung verschieben | - | Investing.com Deutsch | ||
| 09.04. | Theriva Biologics, Inc. - 8-K, Current Report | 1 | SEC Filings | ||
| 23.03. | Theriva gets FDA alignment on phase 3 trial design for VCN-01 | 3 | Investing.com | ||
| 23.03. | Theriva Biologics, Inc. - 8-K, Current Report | 4 | SEC Filings | ||
| 23.03. | Theriva Biologics, Inc.: Theriva Biologics Announces Positive End-of-Phase 2 Meeting with U.S. FDA Regarding the Design of a Phase 3 Trial of VCN-01 in Metastatic Pancreatic Ductal Adenocarcinoma | 501 | GlobeNewswire (Europe) | - Successful meeting with FDA enables advancement into proposed Phase 3 clinical trial of VCN-01 in combination with gemcitabine/nab-paclitaxel for the first-line treatment of metastatic PDAC -
-... ► Artikel lesen | |
| 12.03. | Theriva Biologics GAAP EPS of -$2.08 | 2 | Seeking Alpha | ||
| 12.03. | Theriva Biologics, Inc. - 10-K, Annual Report | 3 | SEC Filings | ||
| 12.03. | Theriva Biologics, Inc. - 8-K, Current Report | 3 | SEC Filings | ||
| 12.03. | Theriva Biologics, Inc.: Theriva Biologics Reports Full-Year 2025 Operational Highlights and Financial Results | 481 | GlobeNewswire (Europe) | - Advancing clinical development of VCN-01 for pancreatic ductal adenocarcinoma and retinoblastoma -
- Licensed SYN-020 to Rasayana Therapeutics for development in multiple indications; up to $38... ► Artikel lesen | |
| 20.02. | Weekly Buzz: NRx Pharmaceuticals Sets The Path For NRX-100's NDA; Disc Medicine Gets FDA's CRL; Theriva Biologics Licenses SYN-020 | 1.370 | AFX News | NEW BRUNSWICK (dpa-AFX) - The biotech space this week witnessed significant key milestones, including FDA approvals, NDA path-setting meetings, rejections, licensing agreements, and oncology... ► Artikel lesen | |
| 18.02. | Theriva Biologics, Inc.: Theriva Biologics Licenses SYN-020 to Rasayana Therapeutics for Multiple Indications | 625 | GlobeNewswire (Europe) | - Theriva received a $300,000 upfront payment at signing and is eligible for up to $38M in development, regulatory, and sales milestones along with tiered single-digit royalties on net product sales... ► Artikel lesen |
1 2 Weiter >>
36 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BAYER | 35,490 | -0,56 % | Börsenrotation ohne KI! Jetzt 100 % mit MustGrow, Bayer und Novo Nordisk, Achtung bei K+S | Erstellt und veröffentlicht im Auftrag von MustGrow Biologics Corp. Perspektivenwechsel bleibt das Gebot der Stunde. Denn gestern tagte die EZB zur aktuellen Lage bei Inflation und Zinsniveau. Mit einer... ► Artikel lesen | |
| NOVO NORDISK | 36,320 | +1,75 % | Warum Novo nordisk heute im Fokus erfahrener Anleger steht: Warum Insider bereits massiv kaufen - Was ich meinen Lesern jetzt rate | ||
| PFIZER | 22,350 | +0,61 % | Rendite-Chance Krebsmarkt: Mit BioNTech, Vidac Pharma und Pfizer den nächsten Mrd.-Trigger erwischen | Die Alterung der Gesellschaft lässt die Gesundheitsausgaben rasant steigen. Im Krebssegment zeichnet sich ein Billionenmarkt ab, denn bis 2040 werden jährlich über 30 Mio. Neuerkrankungen erwartet.... ► Artikel lesen | |
| NOVARTIS | 129,58 | +2,26 % | JEFFERIES stuft NOVARTIS AG auf 'Hold' | NEW YORK (dpa-AFX Analyser) - Das Analysehaus Jefferies hat die Einstufung für Novartis mit einem Kursziel von 110 Franken auf "Hold" belassen. Der Entwicklungschef habe sich sehr positiv zum Sicherheitsprofil... ► Artikel lesen | |
| MERCK KGAA | 138,15 | +0,51 % | EQS-PVR: Merck KGaA: Veröffentlichung gemäß § 40 Abs. 1 WpHG mit dem Ziel der europaweiten Verbreitung | EQS Stimmrechtsmitteilung: Merck KGaA
Merck KGaA: Veröffentlichung gemäß § 40 Abs. 1 WpHG mit dem Ziel der europaweiten Verbreitung
08.06.2026 / 16:57 CET/CEST
Veröffentlichung... ► Artikel lesen | |
| GILEAD SCIENCES | 108,58 | -2,29 % | Neue CAR-T-Erfolge bei Autoimmunerkrankungen: Warum Lösungen für Zelltherapien vor dem nächsten Wachstumsschub stehen könnten | ||
| AURORA CANNABIS | 2,966 | +0,14 % | Aurora Deepens its Impact for Veterans Across Canada | ||
| SANOFI | 77,47 | +1,83 % | JEFFERIES stuft SANOFI auf 'Buy' | NEW YORK (dpa-AFX Analyser) - Das Analysehaus Jefferies hat die Einstufung für Sanofi mit einem Kursziel von 100 Euro auf "Buy" belassen. Bei den Franzosen gebe es trotz des Führungswechsels keinen... ► Artikel lesen | |
| GSK | 22,080 | -0,09 % | DEUTSCHE BANK RESEARCH stuft GSK auf 'Hold' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat die Einstufung für GSK mit einem Kursziel von 1900 Pence auf "Hold" belassen. Emmanuel Papadakis bezeichnete in seinem am Dienstag vorliegenden... ► Artikel lesen | |
| ABBVIE | 195,80 | +1,24 % | Drei Qualitätswerte im Fokus: Was der "Quant Growth And Income"-Ansatz jetzt über Microsoft, Visa und AbbVie verrät | Microsoft, Visa und AbbVie stehen im Zentrum eines quantitativen Wachstums- und Einkommensansatzes, den Seeking Alpha aktuell transparent macht. Der Einblick in das "Quant Growth And Income"-Portfolio... ► Artikel lesen | |
| ROCHE | 344,58 | -1,48 % | JEFFERIES stuft ROCHE HOLDINGS AG auf 'Underperform' | NEW YORK (dpa-AFX Analyser) - Das Analysehaus Jefferies hat die Einstufung für Roche mit einem Kursziel von 230 Franken auf "Underperform" belassen. Das Risiko für die mittelfristigen Ziele der Schweizer... ► Artikel lesen | |
| CANOPY GROWTH | 0,887 | -0,20 % | Canopy Growth Just Made a Big Acquisition: Game-Changer -- or Another Misstep? | ||
| STADA ARZNEIMITTEL | - | - | Enzymatica AB: Quarterly report Q1/2026 - Enzymatica strengthens international position through STADA agreement | First quarter
Net sales amounted to SEK 11.4 million (12.3).
Operating result amounted to SEK -13.6.million (-18.0)
Earnings per share, before... ► Artikel lesen | |
| ELI LILLY | 993,30 | -0,29 % | Eli Lilly: Es geht Schlag auf Schlag - nächster Deal mit Camurus | Der US-amerikanische Pharma-Riese Eli Lilly bleibt auf Shopping-Tour. Neben zwei Lizenzdeals in Asien konnte das Unternehmen auch eine erweiterte Partnerschaft mit der schwedischen Camurus einfädeln.... ► Artikel lesen | |
| ASTRAZENECA | 158,20 | +0,48 % | JEFFERIES stuft ASTRAZENECA auf 'Buy' | NEW YORK (dpa-AFX Broker) - Das Analysehaus Jefferies hat die Einstufung für Astrazeneca mit einem Kursziel von 18.000 Pence auf "Buy" belassen. Die Studiendaten des GLP-1-Abnehmmittels Elecoglipron... ► Artikel lesen |
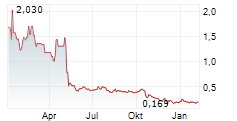
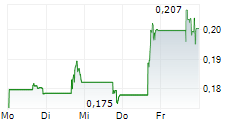
THERIVA BIOLOGICS INC gehört der Branche Pharma an. Der aktuelle Kurs liegt bei 0,239 Euro und damit -10,08 % im Minus.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu THERIVA BIOLOGICS INC finden Sie auf Wallstreet Online.