| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 1,460 | 1,550 | 06.06. |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 22.05. | Innate Pharma SA - 6-K, Report of foreign issuer | 3 | SEC Filings | ||
| 22.05. | Innate Pharma SA: Outcome of Innate Pharma's 2026 Annual General Meeting | 303 | Business Wire | Regulatory News:
Innate Pharma SA (Euronext Paris: IPH; Nasdaq: IPHA) ("Innate" or the "Company") announced the voting results of its shareholders at the Annual General Meeting ("AGM"), which took... ► Artikel lesen | |
| 19.05. | Innate Pharma SA: Innate Pharma to participate in the Jefferies Global Healthcare Conference | 353 | Business Wire | Regulatory News:
Innate Pharma SA (Euronext Paris: IPH; Nasdaq: IPHA) ("Innate" or the "Company"), a clinical-stage biotechnology company developing immunotherapies for cancer patients, today announced... ► Artikel lesen | |
| INNATE PHARMA SA ADR Aktie jetzt für 0€ handeln | |||||
| 16.05. | Innate Pharma übertrifft Erwartungen - Aktie steigt vorbörslich um fast 60 % | 4 | Investing.com Deutsch | ||
| 13.05. | Innate Pharma SA - 6-K, Report of foreign issuer | 4 | SEC Filings | ||
| 13.05. | Innate Pharma reports Q1 results | 8 | Seeking Alpha | ||
| 13.05. | Innate Pharma SA: Innate Pharma Reports First Quarter 2026 Business Update and Financial Results | 495 | Business Wire | Lacutamab TELLOMAK-3 confirmatory Phase 3 trial in cutaneous T-cell lymphoma (CTCL) remains planned for initiation in H2 2026, subject to non-dilutive financing options currently under negotiation... ► Artikel lesen | |
| 12.05. | Innate Pharma SA: Number of Shares and Voting Rights of Innate Pharma as of April 16, 2026 | 295 | Business Wire | Regulatory News:
Pursuant to the article L. 233-8 II of the French "Code de Commerce" and the article 223-16 of the French stock-market authorities (Autorité des Marchés Financiers, or "AMF") General... ► Artikel lesen | |
| 06.05. | Innate Pharma SA - 6-K, Report of foreign issuer | - | SEC Filings | ||
| 06.05. | Innate Pharma SA: Innate Pharma Announces Conference Call and Webcast for Q1 2026 Business Update | 358 | Business Wire | Regulatory News:
Innate Pharma SA (Euronext Paris: IPH; Nasdaq: IPHA) ("Innate" or the "Company"), today announces that the Company will hold a conference call on Wednesday, May 13, 2026 at 1:30... ► Artikel lesen | |
| 23.04. | Innate Pharma SA - 6-K, Report of foreign issuer | - | SEC Filings | ||
| 23.04. | Innate Pharma SA: Innate Pharma to Participate in the D. Boral Capital Global Conference | 320 | Business Wire | Regulatory News:
Innate Pharma SA (Euronext Paris: IPH; Nasdaq: IPHA) ("Innate" or the "Company"), a clinical-stage biotechnology company developing immunotherapies for cancer patients, today announced... ► Artikel lesen | |
| 17.04. | Innate Pharma legt Zwischenergebnisse zu Lungenkrebsstudie vor | 1 | Investing.com Deutsch | ||
| 17.04. | Innate Pharma reports interim lung cancer trial results | 1 | Investing.com | ||
| 17.04. | Innate Pharma SA: Innate Pharma to Present MATISSE Phase 2 Interim Results of IPH5201 in Clinical Trials Plenary Session at AACR 2026 | 525 | Business Wire | Encouraging early results catalyze continued investigation in the MATISSE Phase 2 study evaluating IPH5201, a first-in-class anti-CD39 monoclonal antibody, in combination with durvalumab and... ► Artikel lesen | |
| 13.04. | Innate Pharma SA - 6-K, Report of foreign issuer | - | SEC Filings | ||
| 13.04. | Innate Pharma SA: Number of Shares and Voting Rights of Innate Pharma as of April 9, 2026 | 362 | Business Wire | Regulatory News:
Pursuant to the article L. 233-8 II of the French "Code de Commerce" and the article 223-16 of the French stock-market authorities (Autorité des Marchés Financiers, or "AMF") General... ► Artikel lesen | |
| 13.04. | Innate Pharma SA: Innate Pharma to Hold Its Annual General Meeting of Shareholders on May 21, 2026 | 362 | Business Wire | Regulatory News:
Innate Pharma SA (Euronext Paris: IPH; Nasdaq: IPHA) ("Innate" or the "Company") will hold its Annual General Meeting of shareholders ("AGM") at 10:30 a.m. CEST on May 21, 2026... ► Artikel lesen | |
| 08.04. | Innate Pharma SA: Innate Pharma to Present at the AACR 2026 Oncology Industry Partnering Event | 379 | Business Wire | Regulatory News:
Innate Pharma SA (Euronext Paris: IPH; Nasdaq: IPHA) ("Innate" or the "Company"), announced today that Yannis Morel, Chief Operating Officer, will present at the AACR Oncology... ► Artikel lesen | |
| 03.04. | Innate Pharma SA: Innate Pharma to Participate in the Kempen Life Sciences Conference | 604 | Business Wire | Regulatory News:
Innate Pharma SA (Euronext Paris: IPH; Nasdaq: IPHA) ("Innate" or the "Company"), a clinical-stage biotechnology company developing immunotherapies for cancer patients, today announced... ► Artikel lesen |
1 2 3 Weiter >>
54 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| VALNEVA | 2,331 | -0,64 % | Valneva im Höhenflug: Das Signal das die meisten Anleger noch nicht kennen - Meine Einschätzung für die neue Börsenwoche | ||
| NOVAVAX | 8,080 | -1,70 % | NOVAVAX INC: Stabilität vor der nächsten Bewegung | ||
| OCUGEN | 1,118 | +0,18 % | Ocugen, Inc. - 8-K, Current Report | ||
| INFLARX | 1,594 | -0,44 % | InflaRx NV im freien Fall: Steht die Aktie vor einem Kursdesaster? - Meine Strategie für die kommende Handelswoche! | ||
| INTELLIA THERAPEUTICS | 11,240 | -4,26 % | XETR NEW INSTRUMENTS AVAILABLE ON XETRA - 04.06.2026 | Folgende Wertpapiere werden in den Handel an folgenden Handelsplätzen (MIC Code) im Quotation Board an der Frankfurter Wertpapierbörse (FWB) aufgenommen. Die Notierung an anderen Handelsplätzen im Quotation... ► Artikel lesen | |
| EDITAS MEDICINE | 2,340 | -0,43 % | Editas Medicine, Inc.: Editas Medicine Reports New Preclinical Data Demonstrating Progress of EDIT-401 as Potential Treatment for Hyperlipidemia at the American Society of Gene and Cell Therapy 2026 Annual Meeting | CAMBRIDGE, Mass., May 14, 2026 (GLOBE NEWSWIRE) -- Editas Medicine, Inc. (Nasdaq: EDIT), a pioneering gene editing company developing transformative medicines for serious diseases, shared new preclinical... ► Artikel lesen | |
| VIVORYON THERAPEUTICS | 1,225 | +0,82 % | Vivoryon Therapeutics N.V. Reports Full Year 2025 Financial Results and Provides Business Update | Vivoryon Therapeutics N.V. Reports Full Year 2025 Financial Results and Provides Business Update
Compelling kidney function data and meta-analysis from two Phase 2 studies with varoglutamstat presented... ► Artikel lesen | |
| BIOCRYST PHARMACEUTICALS | 7,232 | -0,85 % | BioCryst Ireland, a Neopharmed Gentili company: Hereditary angioedema, a European initiative to shine a spotlight on a still under-recognized condition | Ahead of World HAE Day, BioCryst Ireland, a Neopharmed Gentili company, organised an international event in Milan bringing together leading experts from across Europe to raise awareness of... ► Artikel lesen | |
| SAREPTA THERAPEUTICS | 13,800 | +0,88 % | Improving Profitability Supports Sarepta Therapeutics Inc. (SRPT) as One of the Most Shorted Small-Cap Stocks to Buy | ||
| PACIFIC BIOSCIENCES OF CALIFORNIA | 1,235 | -1,56 % | PACIFIC BIOSCIENCES OF CALIFORNIA, INC. - 8-K, Current Report | ||
| CARDIOL THERAPEUTICS | 0,989 | +2,17 % | Original-Research: Cardiol Therapeutics Inc (von First Berlin Equity Research GmbH): BUY | Original-Research: Cardiol Therapeutics Inc - from First Berlin Equity Research GmbH
30.04.2026 / 13:07 CET/CEST
Dissemination of a Research, transmitted by EQS News - a service of EQS Group.
The... ► Artikel lesen | |
| GERON | 1,039 | +1,86 % | Geron Corporation Reports First Quarter 2026 Financial Results and Recent Business Highlights | Achieved $51.8 million in RYTELO- (imetelstat) net product revenue in Q1 2026, an increase of 8% compared to the fourth quarter 2025 Reiterated 2026 RYTELO net product revenue and total operating... ► Artikel lesen | |
| ARBUTUS BIOPHARMA | 3,690 | +0,27 % | Arbutus Biopharma Corp - S-8, Securities to be offered to employees in employee benefit plans | ||
| IMMUNITYBIO | 6,070 | +1,17 % | ImmunityBio Inc. (IBRX): One of the Most Shorted Mid-Cap Stocks to Buy Now | ||
| MICROBOT MEDICAL | 1,695 | +5,61 % | Lucid Capital Markets initiates Microbot Medical stock with buy rating |
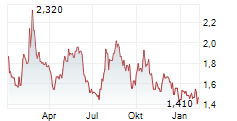
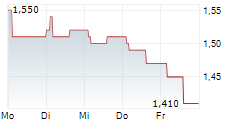
Das Unternehmen INNATE PHARMA SA ADR kann der Branche Biotechnologie zugeordnet werden. Die relative Kursveränderung beträgt aktuell +1,41 % bei einem Aktienkurs von 1,440 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu INNATE PHARMA SA ADR finden Sie auf Wallstreet Online.